Reports
Health
Wellness
Postpartum depression (PPD) is a condition that affects new mothers (or biological mothers) after childbirth.
About 10–15% of adult mothers yearly are affected by it every year.
Symptoms can include feelings of sadness, emptiness, hopelessness, and worthlessness and can last for weeks or months.
It can make it very difficult for new mothers to care for their babies and lead to sleep, appetite, and mood problems.
The effects on children may include behavioral, developmental, socioemotional, and cognitive delay and may last years beyond infancy.
Studies suggest that individual susceptibility to the development of PPD is related to the presence or absence of certain genetic changes associated with PPD risk.
Till now, hormones have been taught to be the biggest contributor to PPD. Estrogen and progesterone levels increase 10-fold during pregnancy.
The dramatic drop in these hormone levels after childbirth can contribute to the symptoms of PPD.
The levels of thyroid hormones seem to play a role in PPD.
They may also drop after giving birth, contributing to PPD.
Sleep deprivation and early-stage motherhood go hand-in-hand.
This accumulated fatigue can cause a dip in mood and energy.
The symptoms of sleep deprivation and PPD have a lot of overlap that sometimes, it can be difficult to each other apart.
The relationship between sleep and PPD is a two-way street; each can aggravate the other.
According to a study, the risk of depression in women with poor sleep quality was 3.34 times higher than in those with good sleep quality.
Compared to mothers with temporary sleep disturbances, those who consistently report very poor or drastically decreasing sleep quality are more likely to develop PPD.
Other risk factors for PPD are:
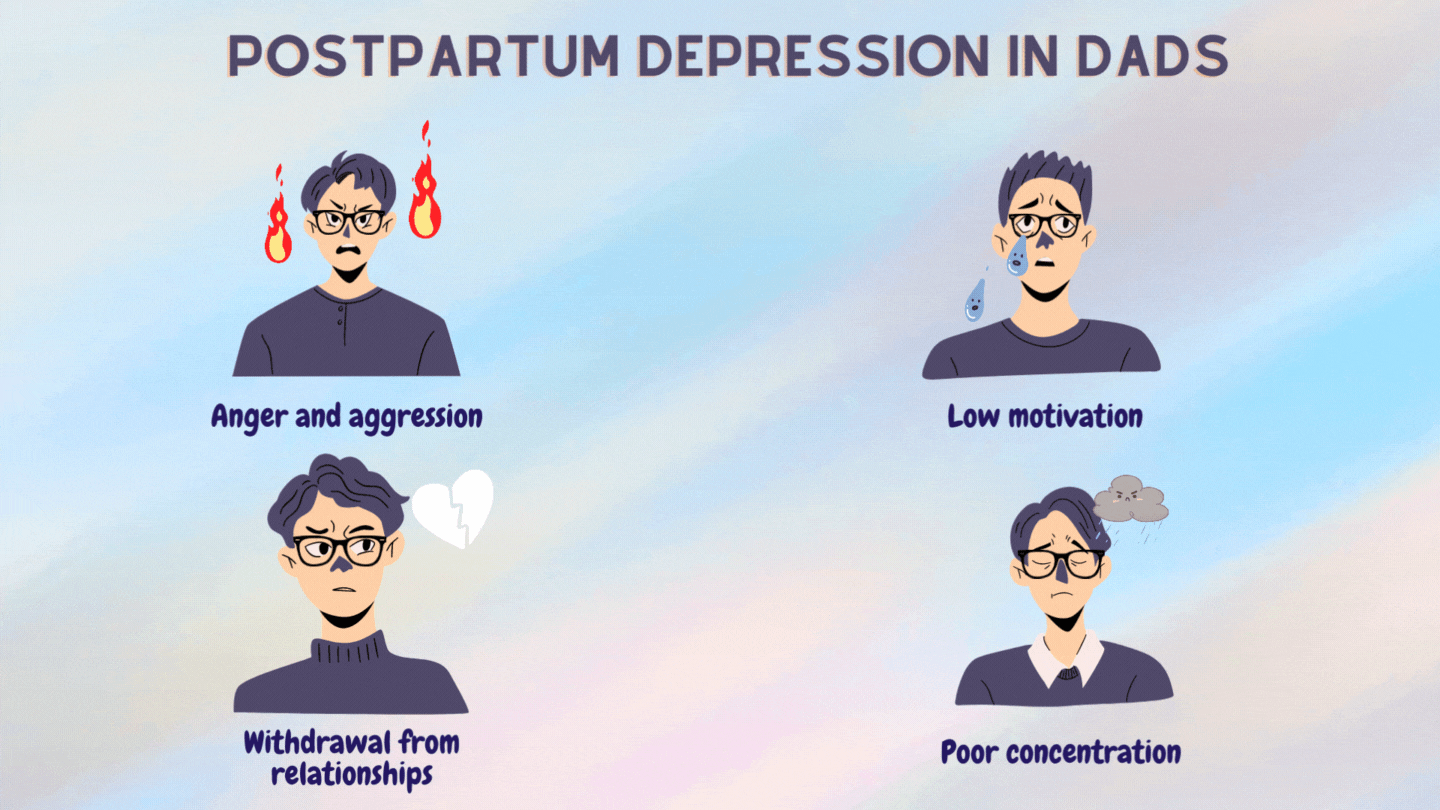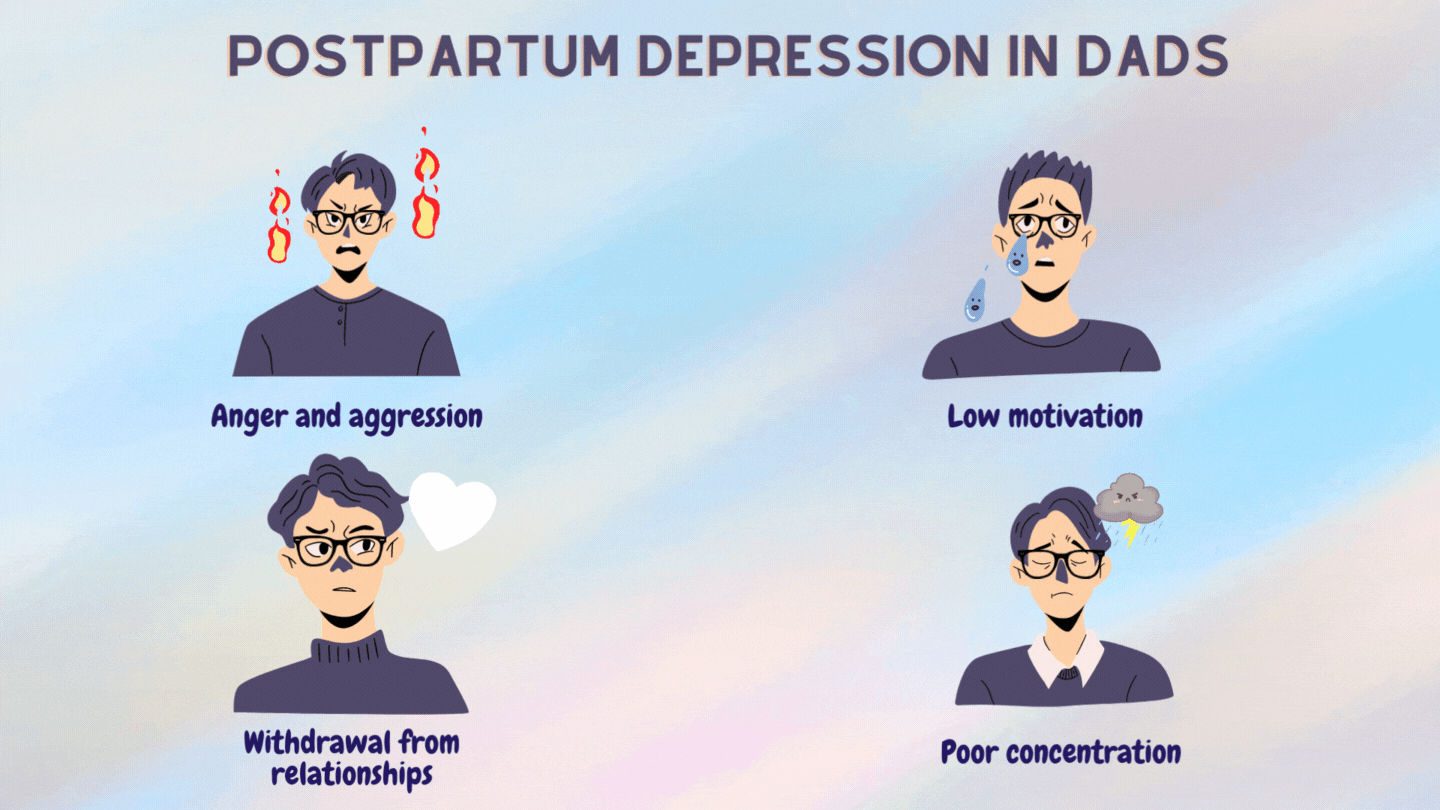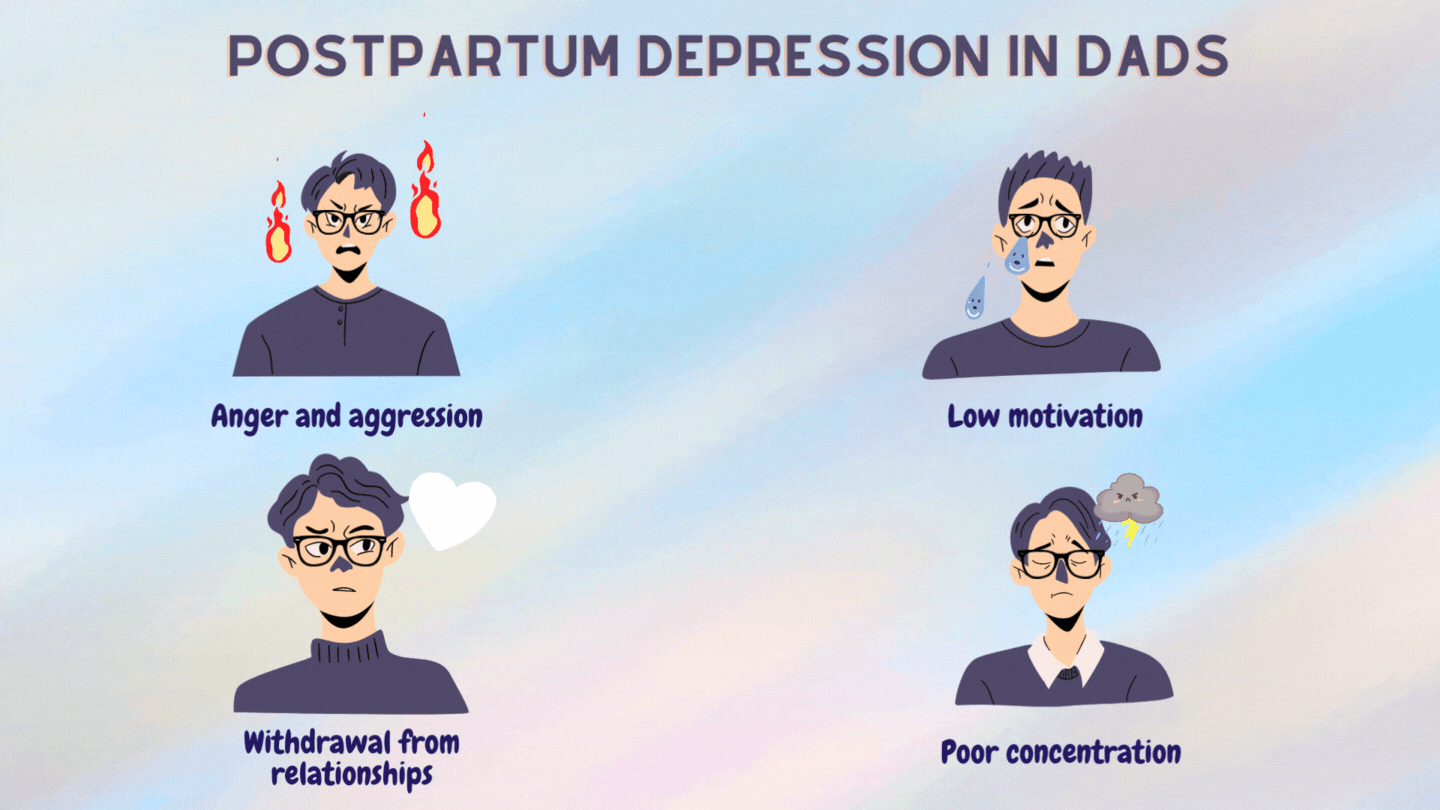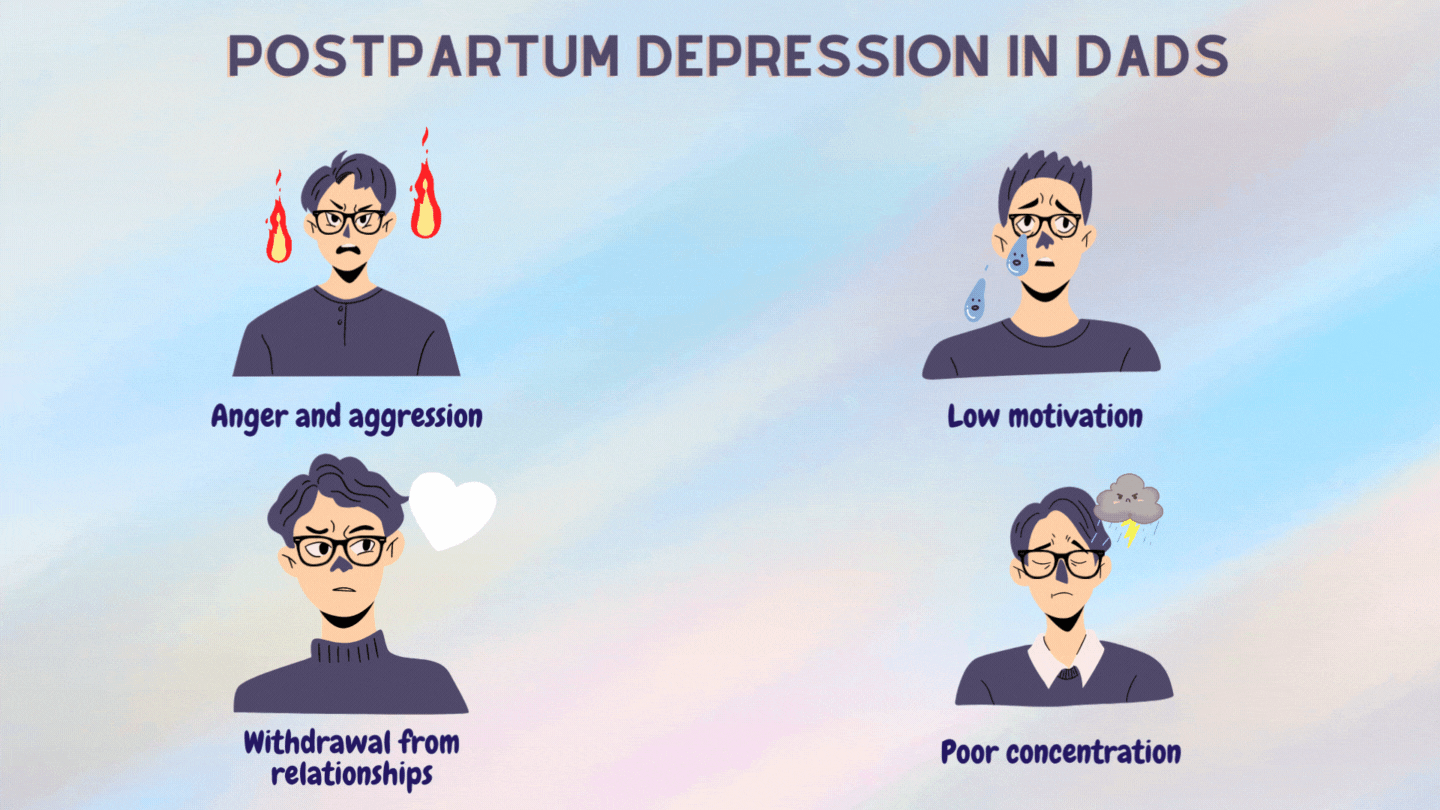
Studies show that 1 in 10 dads suffer from PPD.
Research describes certain hormonal changes in dads that promote their attachment to their newborns.
These may include decreased testosterone levels and increased estrogen, prolactin, and cortisol levels.
The hormonal changes may also increase the risk of PPD in men.
Other non-hormonal factors that are involved in PPD include:
Some signs of PPD in men are:
PPD was long thought to be due to the sudden drop in hormones following childbirth.
However, studies have reported nos significant differences in the hormone levels between depressed and non-depressed mothers.
But there’s a possibility that some may be more sensitive to hormonal fluctuations than others.
A group of researchers from Johns Hopkins studied the epigenetic changes introduced in the hippocampus by estrogen.
*epigenetic changes are DNA modifications that turn a gene “on” or “off.”
*hippocampus is a part of the brain governing moods.
They identified the involvement of two genes, TTC9B and HP1B3, in hippocampal activity.
They may regulate the brain’s ability to recognize and adapt to new situations - the two key elements of mood.
Estrogen seems to behave as an antidepressant, supporting the activity of these genes.
The researchers later replicated this experiment in 52 pregnant women with mood disorders.
They note that those with PPD had significant epigenetic changes in those genes that interact the most with estrogen.
This may make them more sensitive to the hormone’s effects.
TTC9B and HP1B3 genes, specifically, were highly correlated with PPD and predicted with 85% certainty about which women got PPD.
Depression is one of the most common mental disorders all over the world. According to experts, one in five people worldwide will experience a major depressive disorder at least once in their lifetime.
In 2020, 14.8 million U.S adults had at least one major depressive disorder that led to the inability to carry out everyday activities.
The brain uses various chemicals to send and receive information.
These chemical messengers are called neurotransmitters.
When there is an imbalance in brain chemical levels, it could lead to multiple mental health problems, including depression.
According to experts, imbalances in three primary monoamine neurotransmitters, dopamine, serotonin, and norepinephrine, can lead to depressive disorders.
Monoamine neurotransmitters are those chemicals with a single amino group.
These are mostly involved in processing emotions, memories, and arousals.
Here are the specific roles of these three neurotransmitters.
A study reported that depression is a combination of two events - loss of a favorable condition and the presence of an adverse situation.
Imbalances in these neurotransmitters can cause a combination of both these events, eventually leading to depression.
Several reasons can cause imbalances in neurotransmitters. Some common ones include:
Genetically, some people may have increased levels of Monoamine Oxidase A (MOA) in the body.
MOA is an enzyme controlled by the MAOA gene.
Higher levels of MOA break down the neurotransmitters and lead to deficient levels of dopamine, serotonin, and norepinephrine in the body.
There are many nutrients needed for producing and regulating neurotransmitters in the body.
Some of them include:
Nutritional deficiencies can hence reduce the levels of neurotransmitters in the body, leading to imbalances.
Lifestyle choices could be another common reason people have serotonin, dopamine, and norepinephrine imbalances.
For instance, chronic sleep deprivation may cause lower serotonin levels, leading to mental health conditions like depression.
Obesity and excessive consumption of sugar are both associated with dopamine deficiency.
Lack of physical activity is also associated with neurotransmitter imbalances.
Some studies suggest that drug abuse during early developmental periods or psychological stress can act as environmental factors, affecting neurotransmitter levels in the body.
Air pollutants may affect neurotransmitter levels in the body, especially serotonin and dopamine.
A study that analyzed suicidal trends in adolescents concluded that suicidal rates were highest when the pollen counts were high.
Image: Causes of Neurotransmitter Imbalance
Get your vitamin and mineral levels checked, and talk to your doctor about supplementing on nutrients that your body may need. Build your diet plan to include all the micro and macronutrients and choose fresher, healthier ingredients to cook with.
A 2015 study suggests that mind-body therapies like yoga and meditation may help handle neurotransmitter imbalances and, as a result, help combat issues like anxiety and depression.
Stress could cause neurotransmitter imbalances in the brain; mind-body therapies can reduce stress and relax the nervous system.
Studies claim that lack of sleep alters neurotransmitter functions in the brain and leads to changes in the production of chemicals like serotonin and dopamine.
Make sure you sleep at least 7-8 hours a day in a comfortable, dark space that allows the secretion of these chemicals.
If you have problems sleeping well, consult your doctor and get help.
Exercising helps release neurotransmitters like dopamine and endorphin.
Exercise also seems to help get better oxygen supply to the brain, allowing the neurons to transmit and receive signals better.
Exercise is a stress reliever, which may help handle norepinephrine imbalances.
Genetic testing may tell you if you are genetically prone to developing neurotransmitter imbalances, thereby having a higher risk for mental health conditions like depression.
Your doctor may be able to help with preventive therapies and solutions to handle the situation, if so.
If you think you show signs of depression, then the first thing to do is get professional help.
Pharmacogenetics is the study of how certain changes in the genes influence drug responses.
This can be due to variations in the genes that produce proteins or enzymes involved in drug metabolism or interaction.
This field of research is increasingly important as it allows for personalized treatment based on an individual's genetic makeup.
Why personalized treatment?
We all are similar, but of course, we are also different.
And the idea of a "one size fits all" approach to diagnostics and treatment is very flawed.
Different people respond to the same drug differently.
Because of the aforementioned genetic variants, some people may be at increased risk for adverse drug reactions (ADRs) for certain medications.
Others may not experience the desired effect of the drug because they clear it from their body too quickly.
Pharmacogenetic information can help identify medications best suited for a particular person.
It can also factor in when clinicians choose the appropriate dosage for individual patients.
In clinical trials, pharmacogenomics can help identify new targets for drug development.
The benefits of knowing how you'll respond to a medication or which drug will work best for you before you even consume it are not hard to imagine.
One of the popular choices of mental health providers for treating anxiety is psychiatric medications.
Implementing pharmacogenomics here lowers the room for trial and error.
It can also help discover what drugs could potentially cause ADRs in an individual.
The pharmacogenomic approach also helps lower the economic burden as less, or no money will be wasted on drugs that aren't effective for an individual.
Some key players in anxiety medications fall into the following categories:
Serotonin Reuptake Inhibitors (SSRIs): SSRIs, originally designed to treat depression, have revolutionized the treatment of anxiety. They work by blocking serotonin (happy hormone) reuptake, thereby increasing its availability for nerve signaling.
Changes in the CYP2C19 gene link to differences in the metabolism of SSRIs.
People who are ultrarapid metabolizers may not experience the therapeutic effects of the drugs due to their quick elimination from the body.
In contrast, poor metabolizers may have a build-up of the medication in the body and experience side effects, some of which may be adverse.
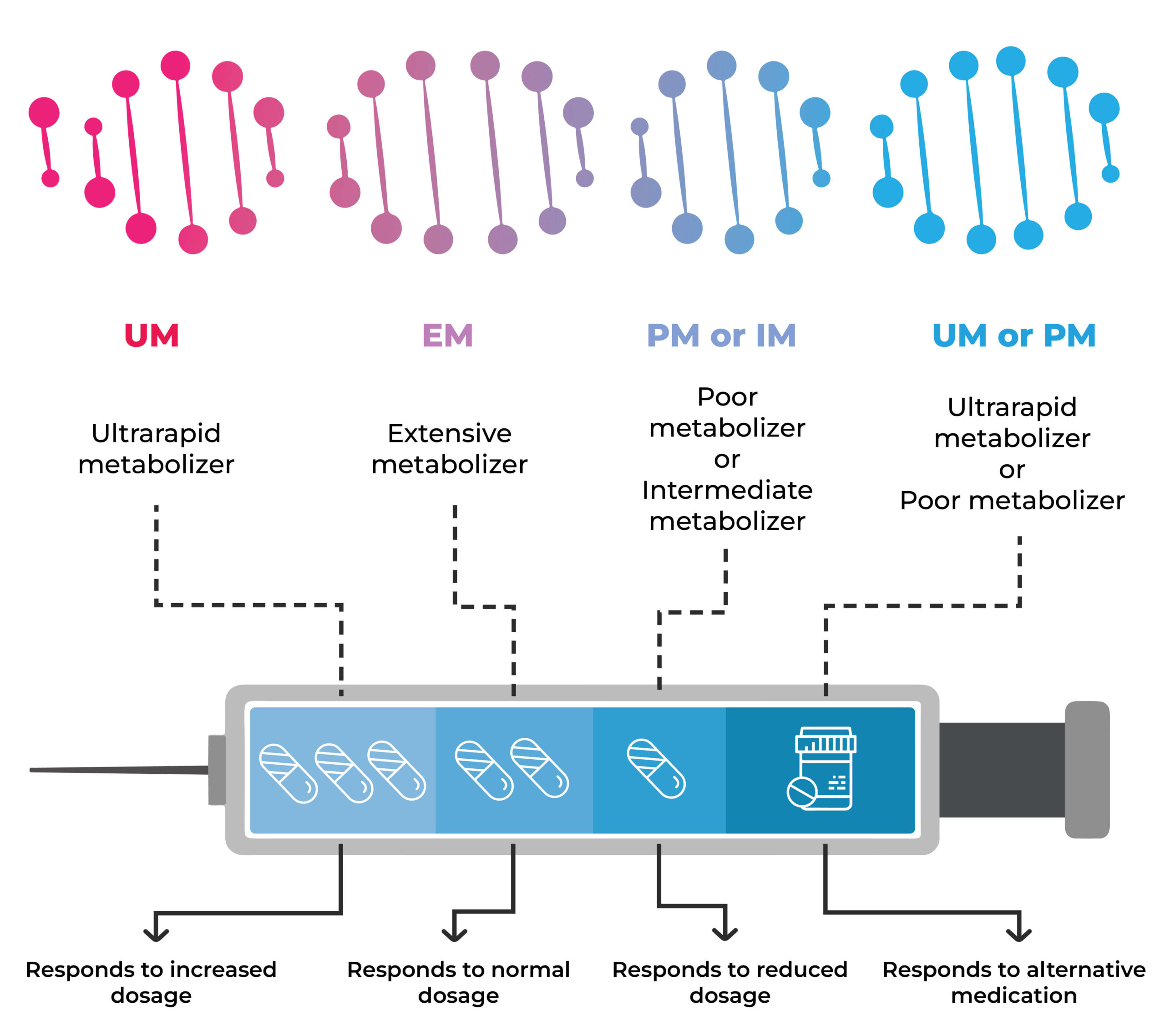
Tricyclic Antidepressants (TCAs): Physicians use TCAs to treat panic disorder, PTSD, anxiety, and depression that occurs with anxiety. They act on 5 brain chemicals (or neurotransmitters). They block the reuptake of serotonin and norepinephrine.
Changes in the CYP2D6 and CYP2C19 genes affect TCAs' exposure time, effectiveness, and safety.
Other medicines used to treat anxiety, like benzodiazepines and serotonin norepinephrine reuptake inhibitors (SNRIs), also have a pharmacogenomic aspect.
Having this information in hand provides many benefits.
Genetic testing for psychiatric medications is a pretty straightforward procedure.
The DNA raw data (the particulars of your DNA in text format) is obtained through a saliva or cheek swab sample.
The raw data is then matched against a database that contains information about the gene changes influencing drug metabolism.
For each drug, the sample DNA data is referenced against the database, and depending on the genetic changes, the metabolizer classification (slow, moderate, normal. Rapid, ultrarapid) is entered.
This report can then be interpreted with the guidance of a qualified medical practitioner and used to understand which drug may work the best for you.
The overall efficacy of genetic testing has improved by leaps and bounds over the years.
However, no genetic test for psychiatric medications has a 100% effectiveness rate.
Genetic testing for medication can be quite expensive.
Studies have found that people would be more inclined to have the test done if the costs were lower or covered by insurance.
A 2017 survey conducted in Singapore reported cost as the biggest concern for pharmacogenomic testing, with over 94% of respondents concerned about cost.
The main concern for mental health practitioners about implementing genetic testing for psychiatric medications is the lack of pharmacogenomic knowledge.
In a survey conducted on psychiatric pharmacists in the US (2015), half of them reported being completely or partially unaware of pharmacogenomics.
Another main concern in employing pharmacogenomics in clinical practice seems to be a lack of clear guidelines about using genetic test results in prescribing psychiatric medications.
Currently, most reports adopt a “traffic light” style for displaying the results.
This leads to a wrong interpretation of ‘green meaning the drug will work’ and ‘red, the drug won't.’
Both clinicians and patients may misinterpret the results.
Anxiety is a normal emotion that may cause you to sweat, feel restless or helpless, and have a rapid heartbeat.
It is a feeling of uneasiness, fear, or dread.
Most people experience anxiety at some point in their lives.
However, if you have anxiety that does not go away or only worsens with time, you may have an anxiety disorder.


There are different types of anxiety disorders, the most common ones being:
Anxiety is caused due to a complex interplay between different factors, including:
Some factors that may increase the risk of developing anxiety are:
Some common symptoms of anxiety disorder are:
Genetics plays a significant role in anxiety and panic disorder.
A family member with anxiety disorder increases one’s risk of developing the condition.
Several studies conducted over the past few decades have reported a genetic association with anxiety. Some key highlights include:
Anxiety does have a genetic component but is influenced by environmental and social factors.
This means that having a family history of anxiety disorder does not confirm you will develop the condition.
It is also possible to develop anxiety disorder without it running in your family.
Monogenic diseases are caused when mutations or abnormal changes in a single gene are inherited by the Mendelian model of inheritance.
A complex interplay between several genes and external factors contributes to the development of anxiety disorders. For this reason, anxiety disorders are said to have a polygenic etiology (caused by multiple genes).
Single nucleotide polymorphisms or SNPs in the following genes have been studied for their association with anxiety disorders:
Those carrying mutations in these genes and subjected to environmental stressors are at higher risk for anxiety than those without the mutations.
Gene-environment interactions emphasize the complex nature of anxiety and its related disorders.
Also Read: Cracking the Anxiety Code Using your DNA Raw Data
Anxiety and anxiety disorders can be treated using medications and counseling.
Treatment options for anxiety include:
The most commonly used medications in the treatment of anxiety disorder are:
This therapy is usually done by a trained psychologist and involves helping you learn about how your emotions affect your behavior.
During psychotherapy, the mental health professional guides you to understand and manage your feelings and fears.
Cognitive-behavioral therapy (CBT) is the most commonly used psychotherapy for anxiety disorder.
Some lifestyle changes to help you manage symptoms of anxiety disorder are:
Anxiety disorders are chronic and cannot be completely cured.
However, with the effective treatment mentioned above, people can manage their symptoms and learn to cope better.
Anxiety is the body’s natural response to stress.
According to the American Psychological Association (APA), anxiety is “an emotion characterized by feelings of tension, worried thoughts and physical changes like increased blood pressure.”
Though we all go through anxiety multiple times in our lives, experiencing persistent and overwhelming anxiety can be debilitating.
When anxiety interferes with your daily activities or causes distress, you may have an anxiety disorder.
Anxiety disorders are mental health disorders, and their cause may be complicated.
Some possible causes:
Studies have shown that anxiety runs in families.
Children of parents with anxiety disorder are seven times more likely to develop the condition.
Though genetics is only 30-40% responsible for anxiety disorder, a recent study on twins showed a direct transmission of anxiety genes from the parent to the child.
Studies also show that over-controlling and anxious parental behavior can promote anxiety in children.
Though genetics play an essential role in the transmission of anxiety in families, environment and parenting also have a significant impact.
A new study found that brain function resulting from anxiety and depression is inherited.
In a research study, young rhesus monkeys were exposed to mildly stressful situations.
It was found that monkeys stopped moving or vocalizing while their stress hormones shot up.
Shy human children show similar behavior.
In another study, researchers found that nearly 30% variation in early anxiety has been credited to family history.
Further, children with highly anxious temperaments are at a 50% risk of developing mental health disorders later in life.
Researchers have found three brain regions involved in anxiety that were heritable. These include:
It is, therefore, believed that the overactivity of the brain that causes anxiety is inherited from our parents.
Parents with anxiety disorders may have no control over the genes they transfer to their children.
However, since environmental and lifestyle factors play a role in anxiety, early intervention can help prevent anxiety in an individual.
Here are some effective ways to break the cycle of anxiety:
Learn to recognize anxiety in your child
As parents with anxiety, it is important to recognize signs of anxiety and nervousness in your child.
You must also teach your child to identify thoughts that scare them.
Apart from recognizing the fear, children must be taught how to deal with their scary or fearful thoughts.
Share your stories
To alleviate fear and anxiety in children, parents must create a safe and trusted environment.
An effective way to do so is to share your stories of vulnerability, anxiety, or depression. Your child may find comfort in knowing that you understand these feelings.
Create smooth communication channels
An essential pillar of support when a child is going through anxiety is knowing whom to speak to when they feel low or anxious.
Keeping communication channels open with your child and initiating conversations about how they are feeling or how their day went can help reduce their anxiety.
Build their confidence
As parents, fixing a problem for your child is an instinct. However, doing so steals from them an opportunity to use and practice their competence.
An excellent way to alleviate your child’s anxiety is to build their confidence by guiding them in problem-solving.
Genetic test
A genetic test can help understand the risk for anxiety and provide personalized recommendations to help prevent it.
Anxiety is a feeling of worry, fear, and tension affecting about 40 million adults in the United States.
Anxiety can be caused by many things, including life stressors, health problems, genetics, and traumatic events.
Anxiety symptoms include nervousness, restlessness, difficulty concentrating, and muscle tension.
Many people also experience occasional panic attacks, which are sudden episodes of intense fear or anxiety.
The five major types of anxiety disorders are:
Generalized Anxiety Disorder, GAD, is a specific anxiety condition marked by persistently elevated tension, anxious feelings, and negative thoughts, even without a force or occasion to trigger them.
Obsessive-compulsive disorder (OCD) is a persistent pattern of evaluations and ritualistic behaviors.
OCD is characterized by unwanted thoughts (obsessions) and repetitive behaviors (compulsions).
Practicing behaviors such as washing hands, counting, checking, or cleaning is typically done with the aim of eliminating thoughts or making them disappear.
However, these so-called "rituals" are only temporary relief, not a solution.
Panic disorder is an anxiety disorder that causes sudden and repeated episodes of intense fear, accompanied by symptoms including chest discomfort, heart palpitations, shortness of breath, dizziness, or stomach distress.
Post-traumatic Stress Disorder, or PTSD, is an anxiety disorder that can develop after exposure to a terrifying event or ordeal that involves impending physical harm.
Traumatic events that may trigger PTSD include traumas arising from violent personal assault, natural or human-caused disasters, accidents, or military combat.
Social Anxiety Disorder (SAD) or social phobia is an anxiety disorder marked by excessive self-consciousness in social situations and overwhelming anxiety.
Depending on the severity of its symptoms, SAD can be limited to only one type of situation, such as a fear of speaking in formal or informal settings.
It also may be so severe that the person may experience the symptoms by simply being around other people.
In the past, scientists believed that a marble-sized brain area, called the amygdala, was responsible for fear and anxiety.
Some studies have shown monkeys with damage to the amygdala were unusually stoic when confronted with scary stimuli - like a snake.
Today, we understand that anxiety results from interactions among different brain regions.
A single brain region cannot drive anxiety.
The amygdala is a part of the emotional response center located deep within the brain.
According to a hypothesis, we only feel anxiety when the emotional brain overpowers the cognitive brain and controls our consciousness.
The dorsal anterior cingulate cortex (DACC) is a part of the cerebral cortex designed to heighten fear signals from the amygdala.
When anxious individuals view pictures of frightened faces, the DACC and amygdala (among other locations) increase communication with each other, which leads to intense anxiety.
But people without anxiety show negligible to no response.
On the other hand, a part of the frontal lobe called the ventromedial prefrontal cortex serves to reduce anxiety signals from the amygdala.
People with damage to this portion of the brain are more inclined to experience anxiety since there's no dampening of the anxiety signals.
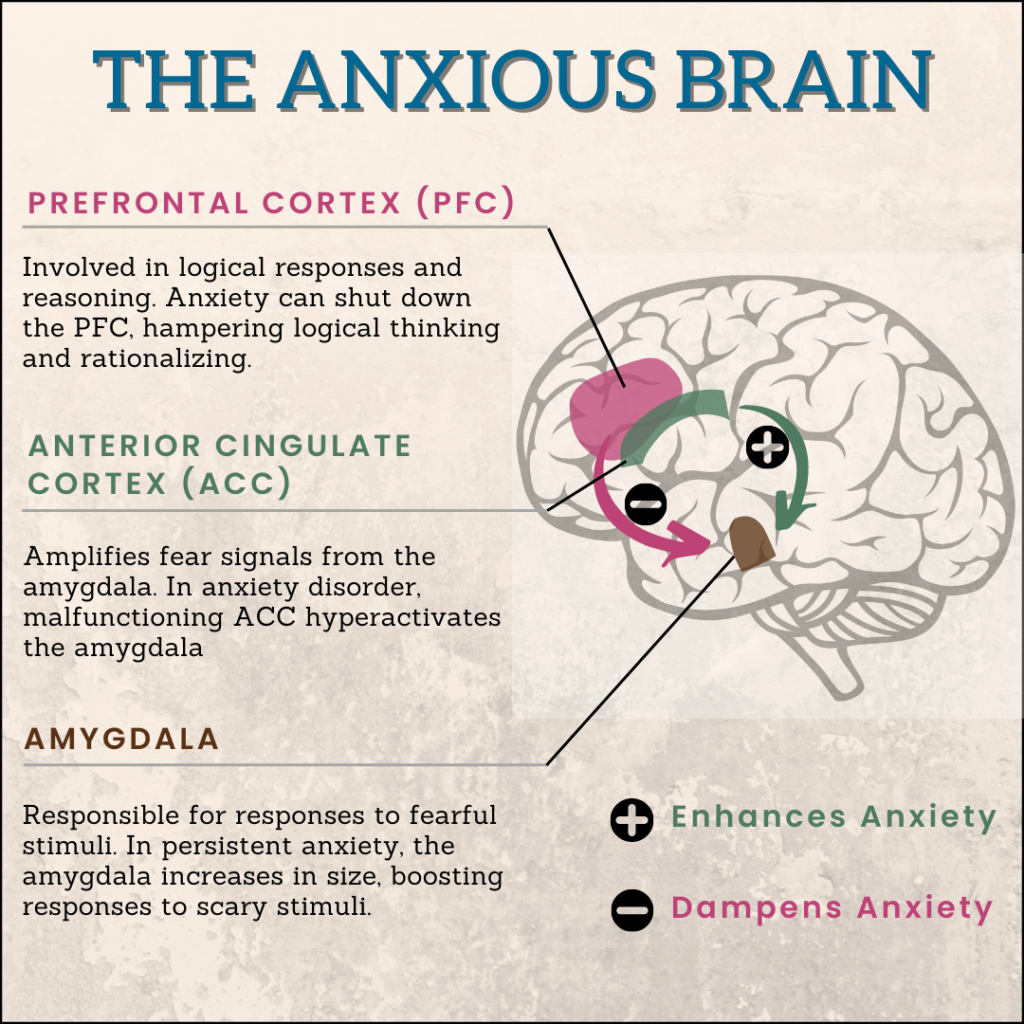
Image: The Effects of Anxiety on the Brain / Inspired by NewScientist
Increases the Levels of Stress Hormones
Anxiety activates the fight or flight response in our bodies.
When fighting off the anxiety trigger(s), the brain releases adrenaline and cortisol, which alerts our bodies and helps us cope with danger.
When this danger is gone, the sympathetic part of the nervous system takes over and brings a sense of calm.
In people suffering from anxiety, the sympathetic nervous system doesn’t do its job well.
As a result, the stress hormones keep getting released, triggering anxiety symptoms.
Increases the Risk of Neuropsychiatric Disorders
Chronic stress and anxiety can lead to structural degeneration and decreased functionality of the hippocampus and the prefrontal cortex.
This can increase the risk for psychiatric disorders, including depression and dementia.
Changes in The Amygdala and Hippocampus
The amygdala is an almond-shaped mass of gray matter in the brain involved with expressing emotions.
It is responsible for responses to fearful and threatening stimuli.
With persistent anxiety, the amygdala increases in size, amplifying the body’s response to scary situations.
The hippocampus plays a major role in learning and memory.
It gets severely affected in mosy psychological disorders, including anxiety.
In chronic anxiety, the hippocampus shrinks in size.
Any damage to it can cause difficulty recalling memories and forming new ones.
Hippocampus is also responsible for linking a particular place to a fearful event.
This explains why some individuals with anxiety may avoid certain places.
Difficulty with Rationalizing
The prefrontal cortex region of the brain is involved in logical responses and reasoning.
It helps process information and makes an informed decision to solve a problem.
The connection between the prefrontal cortex and amygdala is weakened in anxious brains.
This can increase the sensitivity to dangers and hamper logical thinking and rationalizing.
Holding Onto Negative Memories
Hippocampus is responsible for holding onto memories.
With chronic anxiety, the hippocampus shrinks in size, making it difficult to recall memories.
However, anxiety makes the hippocampus think that memories associated with danger and anxiety are good memories to store.
Thus, the memories you manage to recollect may be those of anxiety and stress.
In an anxious brain, the memories of failure, threat, and danger overpower that of success, happiness, and achievement.
Depression
In anxiety, the brain tends to hold onto negative memories.
This contributes to other mental disorders like depression.
While there is no one-size-fits-all approach to coping with anxiety, several strategies can help.
Some of these include:
If needed, seek professional therapy to help address your anxiety.
Gene therapy is a medical approach that treats and prevents genetic and inherited conditions.
Using gene therapy, doctors can treat a condition by altering an individual’s genetic makeup with surgery or medications.
The earliest forms of gene therapy used gene transfer or gene addition, where a new gene or a non-faulty copy of a gene was introduced into the cell to fight disease.
The latest technology revolutionizing gene therapy is CRISPR-Cas9 or genome editing.
Several advanced molecular tools are used in gene-editing and are currently being studied to:
Today, gene therapies are used to treat a few diseases, including Leber congenital amaurosis, spinal muscular atrophy, and autism.
The use of gene therapies for several other conditions is underway.
The main focus of gene therapy is still treating conditions caused by mutations in a single gene.
However, since ASD is not associated with a single gene, using gene therapy to prevent and treat the condition or spectrum of disorders is challenging.
There are two types of ASD:
Caused by abnormal changes or mutations in a single gene and is considered a neurological condition like Fragile X syndrome.
Caused by unknown genetic or environmental factors.
Syndromic ASD is usually more severe, and since mutations of only one gene (monogenic) are responsible for this condition; gene therapy is most promising in these conditions.
Currently, gene therapies that permanently alter the genome are being developed for over 11 monogenic autism syndromes.
Gene therapies for autism depend upon the genetic cause and structural variants of the gene causing the condition.
Accurate diagnosis and precise prediction of the sequence in a mutation will enable the safe application of gene therapy in the future.
A gene called the UBE3A is considered a risk gene for autism.
The UBE3A gene is vital for learning.
When the nerve cells in the brain fire impulses, the activity of the UBE3A gene rises and switches off by itself.
It is this activity that enables us to learn.
When a region in the UBE3A gene is lost or in excess, the learning cycle is affected, resulting in learning difficulties characteristic of people with ASD.
Scientists Zylka and Yi demonstrated that the drug Rolipram suppresses the overactive UBE3A gene.
So, by screening individuals with increased UBE3A gene activity, targeted treatments can be given to alleviate ASD symptoms.
This way, genetic research, and testing can help treat diseases like ASD.
Genetic Testing For Autism: Use Your DNA Raw Data
https://www.iflscience.com/health-and-medicine/can-genetics-find-cure-autism/
https://www.science.org/content/article/gene-therapy-ready-treat-some-forms-autism
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4000534/
Mental health is a topic that is often talked about but remains largely unknown.
When someone says mental health, “genes” or “genetics” is not the next immediate thing that comes to mind, maybe not even the fourth or the fifth thing.
However, recent research into mental health urges us to think otherwise by establishing a significant genetic component for many mental health disorders.
These results can be crucial for understanding and treating mental health disorders.
Thinking about it logically, this shouldn’t surprise us at all!
The millions of activities that go on in the brain contribute to our thoughts, emotions, love, consciousness, and so on.
Millions of brain cells or neurons transmit the brain activities or signals.
So for effective brain activity, the brain cells must function well.
Each cell has DNA with thousands of genes. And if you have “normal” genes, your brain cells function normally.
However, if you carry a “change” or an “error” in one or more of the genes in the DNA of the brain cells, their functioning may be hampered.
Imagine an electricity grid; what’d happen if one circuit is flawed?
It may not result in a complete blackout, but we may be facing power fluctuations.
The brain is only 1000 times more complex! Small changes may not entirely contribute to a mental disorder - there is no one “depression gene” or “anxiety mutation.”
But genetic factors make some people more likely to have mental health issues by interfering with the normal functioning of the brain cells.
This could explain why issues like depression or bipolar disorders seemingly run in the family.
Depression is one of the most common mental illnesses in the United States, with an estimated 15 million Americans affected each year.
A growing body of research suggests that depression may be partly due to genes passed down from family members.
Studies have shown that depression is more common in families with a history of mental illness.
This suggests that there may be a genetic component to depression.
It is estimated that heritability – the proportion of variation in a trait due to genetic factors – accounts for about 40-50% of the risk for developing depression.
Glutamate Receptors and Mood Disorders
Years’ worth of research shows that glutamate is instrumental in depression.
There is also increasing evidence of a possible relationship between glutamate and the pathophysiology and the treatment of depression.
Glutamate is a neurotransmitter (or chemical messenger) released in the brain important for learning and memory.
Excessive release of glutamate in the body may be associated with excitotoxicity-induced brain damage.
Glutamate is synthesized in the body and ingested from certain kinds of food, including meat, cheese, and food seasonings containing MSG or monosodium glutamate.
High levels of glutamate have been linked to mood disorders like depression.
Glutamate receptors play a role in determining the levels of glutamate.
People with certain changes in the gene coding for glutamate receptors may be more prone to depression than others.
Evidence-based studies reveal that anxiety, in part, is dictated by our genes.
Experts noticed a connection between genes and anxiety even before discovering the role of DNA and genes.
If you have an immediate relative with anxiety, there is a 2 to 6 times increased risk of developing it.
The serotonin transporter gene (5-HTT) is one of the well-researched genes for anxiety.
Serotonin is a neurotransmitter or a brain chemical.
It is believed to be a mood stabilizer, which can aid healthy sleep patterns and boost your mood.
The 5-HTT gene is a crucial regulator of serotonin levels in the brain.
A clear correlation has been established between low serotonin levels and increases in depression, anxiety, and other mental health challenges.
The 5-HTT gene comes in two forms: L and S.
The S form is associated with lower 5-HTT gene expression and subsequently lower serotonin re-uptake.
This results in an increased risk for anxiety.
Before starting on the genetic link to stress, it is important to know that stress is not always a villain.
There are two types of stress: eustress and distress,
Most of us know that distress is a negative feeling characterized by extreme sorrow, sadness, and pain - the bad kind of stress.
Eustress is a positive form of stress that produces positive feelings of excitement, fulfillment, meaning, satisfaction, and well-being.
One of the most important genes connected with your stress response is COMT.
The COMT gene produces the catechol-O-methyltransferase or the COMT enzyme.
COMT breaks down neurotransmitters like epinephrine, norepinephrine, and dopamine.
Your fight or flight response is activated in response to excessive stress and is triggered by epinephrine and norepinephrine.
Dopamine plays a crucial role in reward response.
A selected COMT gene modification called rs4680 or V158M has been proven to bring about changes in intellectual and behavioral traits, as well as other illnesses.
People with the GG version of the COMT gene tend to have low dopamine levels and perform better under stress.
Those with the AA version tend to have lower stress resilience.
Xcode Life’s genetic test analyzes genes associated with mental health, like the ones mentioned above.
With the results, you can understand your risk for various mental conditions, including but not limited to depression, anxiety, schizophrenia, stress, and anorexia.
The reports also include customized recommendations to improve your mental health.
Autism or autism spectrum disorder (ASD) is a neurological developmental disability that impacts an individual's communication, behavioral and social skills.
It affects around 1 in 59 children in the United States.
The signs of autism usually appear in early childhood, by 12 to 18 months or earlier.
Since autism is a spectrum disorder, each individual's signs and symptoms vary.
Though there are several types of autism, they are all caused by a combination of genetic and environmental factors.
While people with autism tend to have many challenges, they also are found to be skilled in few activities.
Due to the broad nature of this disorder, autism may occur with other conditions.
Though autism was earlier presumed to be environmental in origin, hundreds of genes are associated with this condition today.
These genes contribute to the various deficits in communication, cognition, and behavior typical in people with autism.
This was proved by a study conducted in the 1970s, which stated that monozygotic twins were more likely to share a diagnosis of autism than dizygotic twins.
Later, several other studies demonstrated autism has a strong genetic component.
Some genes that have been associated with autism include:
Environmental conditions such as maternal immune response in the womb or complications during birth may trigger genetic factors to cause autism.
Research suggests that girls with autism have more mutations than boys with the condition.
Further, studies have shown that boys may inherit the autism gene mutations directly from their unaffected mothers.
This suggests that girls may be more resistant to autism mutations than boys, who are more likely to develop the condition.
Large-scale genome-wide sequencing studies of several psychiatric disorders have revealed a significant and extensive overlap of the risk regions (loci) with autism genes, making it difficult to differentiate between them.
Studies have demonstrated overlap of loci in five major psychiatric disorders (ASD, schizophrenia, bipolar disorder, ADHD, and major depressive disorder) in the following genes:
Recent studies have also found mutations in loci of a few other genes like the dedicator of cytokinesis 8 (DOCK8) and KN motif and ankyrin repair domains 1(KANK1) in all the five conditions.
Fragile X syndrome is one of the most commonly identified causes of inherited intellectual disability and autism or ASD.
However, most people with fragile X syndrome do not have a diagnosis.
Fragile X syndrome derives its name from the microscopic appearance of the X chromosome.
When observed under the microscope, the X chromosome in these individuals appears as it could easily break.
Males receive the X chromosome from their mother and the Y chromosome from their father. Females receive one X chromosome from their mother and one from their father.
So, males inherit the fragile X chromosome only from their mother, but females can inherit it from both parents.
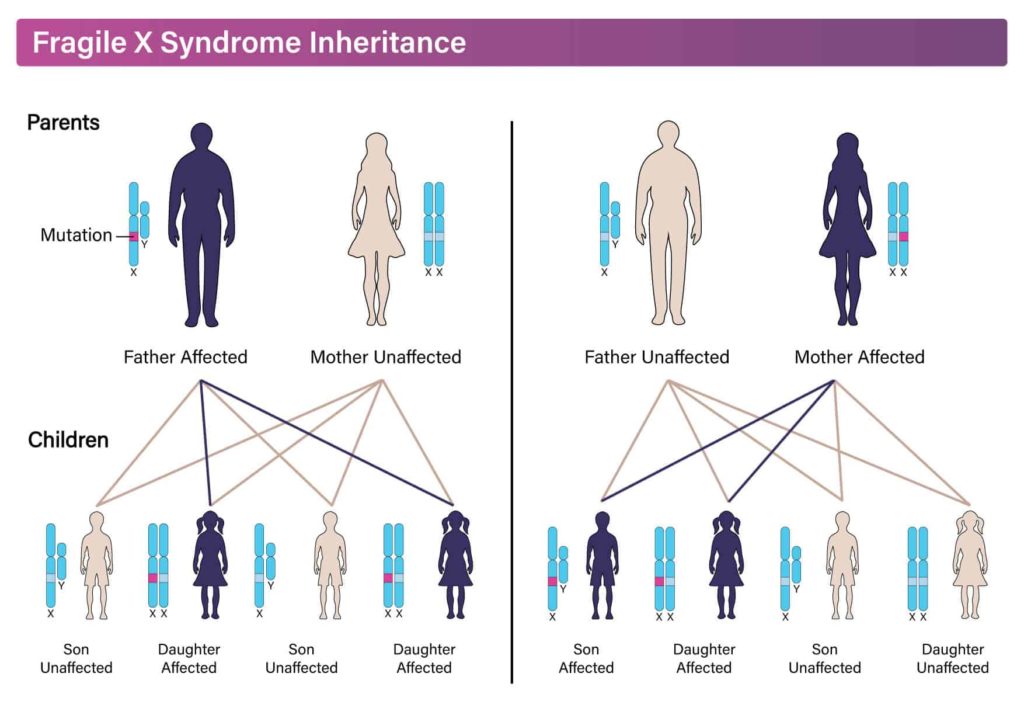
Image: Fragile X Syndrome Inheritance
For this reason, females who have only one fragile X chromosome have protection from the features of the condition and tend to have milder symptoms.
On the other hand, since males have only one X chromosome, they experience more significant learning and emotional disabilities than females.
Around 80% of males with fragile X syndrome show intellectual disability, poor cognitive abilities, emotional problems like anxiety, and obsessive worrying, all of which are similar to autism.
People with fragile X syndrome may also show other features similar to autism, such as
Reference links:
