Reports
Health
Wellness
Caffeine is a psychoactive stimulant consumed worldwide mostly via two beverages, tea, and coffee. Pregnant women have always been advised to limit their caffeine intake since they may metabolize caffeine slowly. Studies also suggest that it can cross the placenta and enter the baby's bloodstream. A recent study published in the International Journal of Epidemiology reported that caffeine consumption alone might not contribute to adverse outcomes in pregnancy.
Pregnant women are often advised to cut off or limit their coffee consumption, owing to the reduced clearance of caffeine during pregnancy.
Pregnant women require a 1.5 to 3.5 times longer half-life to eliminate caffeine than non-pregnant women.
Caffeine, being a stimulant, increases the heart rate and blood pressure, because of which some people may experience jitters.
Further, since caffeine can cross the placenta to enter the baby's bloodstream, it is thought to endanger their health.
The safe upper limit for caffeine consumption in pregnancy is still a bit hazy.
According to the current WHO guidelines, pregnant women should restrict their caffeine intake to less than 300 mg/day (approximately 2-3 cups/day).
In comparison, the World Health Organization (WHO) allows up to 400 mg of caffeine per day for non-pregnant healthy adults.
On the other hand, the NIH advises pregnant women not to consume more than 200 mg of caffeine daily.
Studies on the effects of moderate caffeine consumption (≤200mg/day) report contradictory results.
Some find an increased risk for low body weight at birth and other poor outcomes, while others report no such risk.
Yet another study reported an average of 66 grams of lower birth weights in infants born to mothers who consumed about 50 milligrams of caffeine daily.
But it's important to note that such observational studies may not separate caffeine consumption from other risk factors such as alcohol consumption and smoking.
Thus, the unfavorable outcomes in pregnancies could also be attributed to other risk factors.
The CYP1A2 gene has the largest association with caffeine-related traits as the CYP1A2 enzyme is responsible for 95% of caffeine metabolism.
The CYP1A2 gene exists in different forms in different people.
Depending on the CYP1A2 gene variant one carries, they may metabolize caffeine slowly, at a normal rate, or fast.
This, in turn, impacts a person’s risk for caffeine-associated negative side effects.
Also Read Coffee And CYP1A2: 3 Ways High Caffeine Intake Can Affect You!
AHR is another gene that influences caffeine consumption.
This gene produces an enzyme that induces the activity of the CYP1A2 enzyme.
The more the AHR enzyme, the more the CYP1A2 enzyme, and the more the tendency for increased caffeine consumption.
The ADORA2A gene is associated with the wakefulness-promoting property of caffeine.
It also influences the effect of caffeine on sleep.
The A2A receptor produced by this gene is the main target for caffeine.
Typically, a molecule called adenosine binds to the A2A receptor and helps with sleep regulation.
Upon caffeine consumption, the caffeine molecules displace adenosine molecules from their receptors and sit in their place, promoting wakefulness.
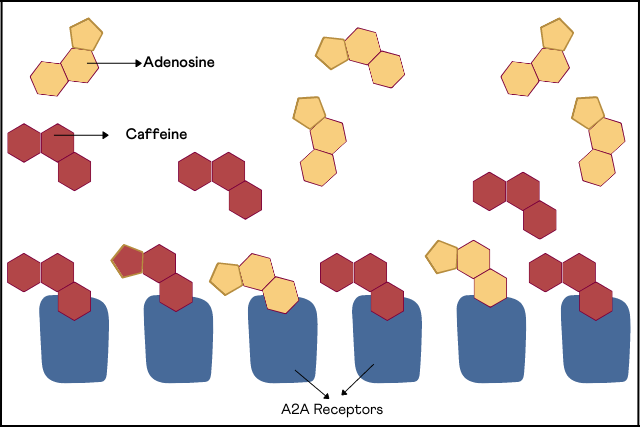
Image: Interaction of Caffeine with A2A Receptors
Certain changes in this gene increase the risk for caffeine-induced insomnia.
The study, published in the International Journal of Epidemiology, investigated a potential causal relationship between caffeine consumption and adverse pregnancy outcomes.
While many previous studies have reported an increased risk of poor pregnancy outcomes with caffeine consumption, it’s been difficult to separate coffee drinking from other risk factors.
The researchers analyzed 8 genetic changes associated with caffeine consumption.
Additionally, they examined whether these changes were associated with birth outcomes (miscarriages, stillbirths, or pre-term birth).
Genome sequencing was performed in individuals who reported these outcomes, and the results were referenced against the controls.
The study reported that there was no greater risk of miscarriage, stillbirth or premature birth for women who drank coffee.
The study looked at the effects of caffeine only in terms of adverse outcomes.
Caffeine could still affect other aspects of fetal development.
So, Dr. Moen, one of the authors of the study, says, "we don't recommend a high intake during pregnancy, but a low or moderate consumption of coffee may be safe."
Certain genetic changes influence caffeine consumption in an individual.
These changes may be why one can consume 7 cups of coffee and still go to bed by 10, and another struggles to sleep even after a couple of cups.
They affect caffeine consumption in both the general population and pregnant women.
The current recommendations for caffeine consumption during pregnancy are based on observation studies where it's difficult to separate coffee drinking from other risk factors like smoking, alcohol, or poor diet.
But caffeine alone doesn't seem to increase the risk for pregnancy-related adverse outcomes.
https://academic.oup.com/ije/advance-article/doi/10.1093/ije/dyac121/6605011#358171758
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4507998/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7209255/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6947650/
Vitamin B12, also known as cobalamin, is a water-soluble vitamin. It is essential for the formation of red blood cells (RBCs), synthesis of genetic material- deoxyribonucleic acid (DNA), and development and function of healthy nerve cells. The deficiency of vitamin B12 can increase the risk of anemia, celiac disease, nephropathy, and immune system disorders. A recent study has reported that diabetic patients using metformin in their treatment are more prone to vitamin B12 deficiency.
Diabetes Mellitus is a chronic disease caused due to the impaired production of insulin by the pancreas, leading to elevated blood glucose levels.
There are three types of diabetes:
Type 1 diabetes
Type 1 diabetes is an autoimmune condition in which the body’s immune system attacks the β-cells of the pancreas.
This results in a lower/no production of insulin by the pancreas, thereby leading to diabetes.
Type 2 diabetes
Type 2 diabetes occurs when the body does not respond appropriately to the insulin hormone secreted by the pancreas.
This leads to the overproduction of insulin by the pancreas, causing weight gain and other complications.
Gestational diabetes
Gestational diabetes is when pregnant women without a history of diabetes suddenly become diabetic during the course of pregnancy.
Long-term usage of metformin has been known to increase the risk of vitamin B12 deficiency by affecting its absorption in the intestine.
A unique protein called intrinsic factor binds to vitamin B12 to facilitate its absorption in the intestine.
Vitamin B12 and protein complex are absorbed through a calcium-dependent membrane.
Metformin impacts the activity of this membrane, subsequently influencing vitamin B12 absorption.
Bile acid (steroid acids produced by the liver) and hepatic cells are essential for vitamin B12 absorption.
Metformin reduces the absorption of bile, resulting in the impaired circulation of vitamin B12 in the liver.
Also Read: Genes And Vitamin B12 (Cyanocobalamine) Needs
The researchers at the Al-Qura University, Saudi Arabia, aimed to estimate the prevalence of vitamin B12 deficiency in type 2 diabetes patients using metformin.
The study results were published in the journal Human Nutrition and Metabolism.
206 patients with type 2 diabetes and metformin were included in the study.
Vitamin B12 serum levels of the patients were estimated.
The patients with serum levels below 243 nmol/L were defined as vitamin B12 deficient.
The patients' medical history, demographics, diet, and metformin use were also assessed.
This study design may not be the best way asses the relationship between metformin usage and vitamin B12 deficiency.
The risk for vitamin B12 deficiency in people on metformin for diabetes is elevated.
This risk is influenced by metformin dosage, duration of metformin use, and occurrence and duration of diabetes.
Routine monitoring of vitamin b12 levels may be needed in people with type 2 diabetes, especially those who have been on 1000 mg/day of metformin for > 2 years.
Check Your Genetic Response to Metformin with the Personalized Medicine Report
Get Nutrition & Diet Insights from Your 23andMe, AncestryDNA Raw Data!
Avocados, being high fiber-dense, play a crucial role in weight loss by suppressing the appetite and promoting metabolic health. A recent study has reported that an avocado a day could help redistribute belly fat in women towards a healthier profile. The study further reported that avocados could also impact the way body fat is stored.
There are two types of abdominal fat in the human body.
The fat present right underneath the skin is called subcutaneous fat.
The fat stored in the deeper layers is called visceral fat.
Image: Types of abdominal fat
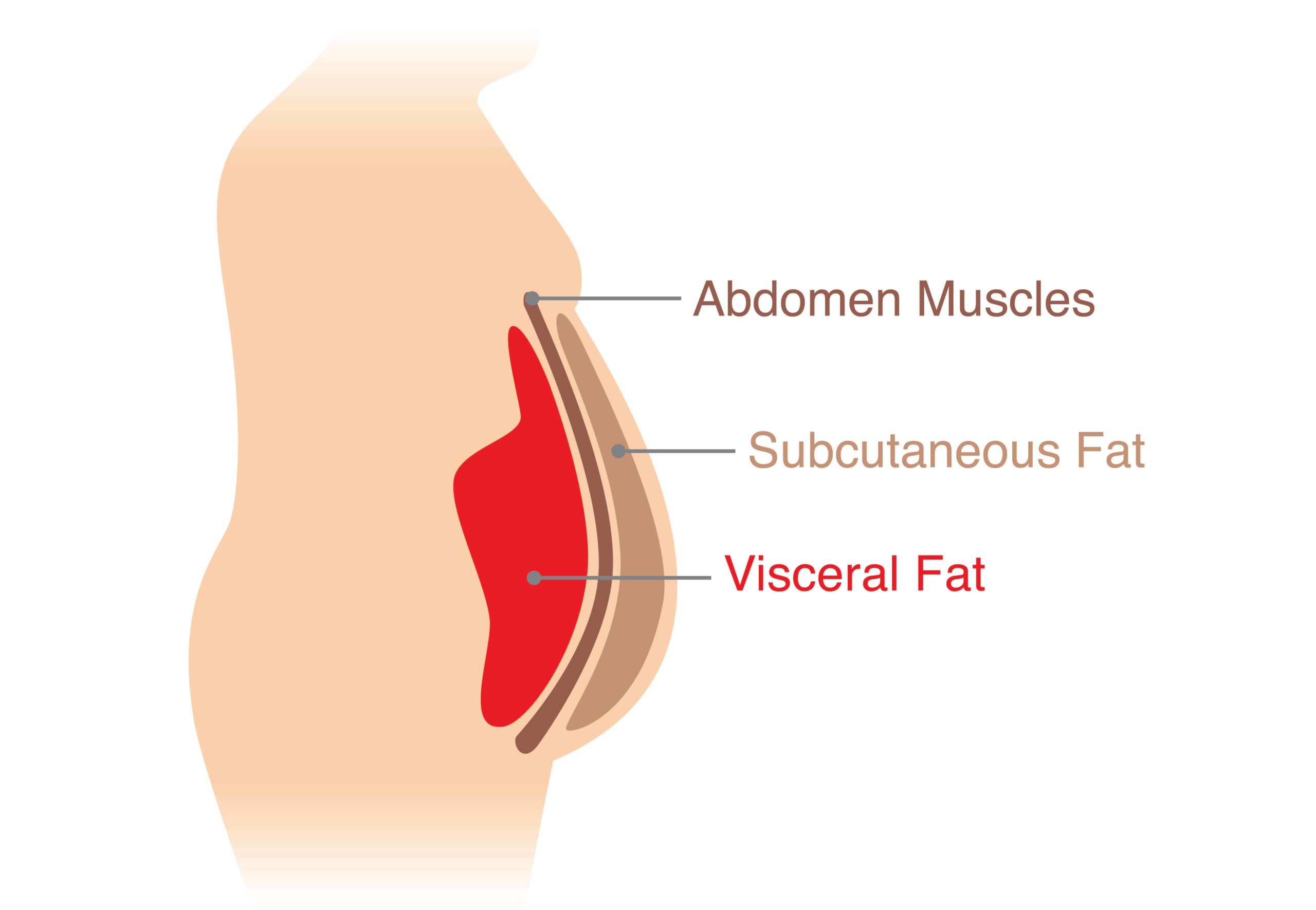
Subcutaneous fat can be pinched, but the visceral fat can only be seen and measured and not pinched.
Several factors are responsible for the accumulation of belly fat, including poor diet, lack of exercise, and stress.
However, improving nutrition, increasing activity, and making other lifestyle changes can help in losing the accumulated fat.
Health complications from subcutaneous fat are more harmful than complications from visceral fat.
Excess belly fat can increase the risk of:
It is very easy to dismiss the weight loss properties of avocados due to their high-fat content.
However, the fact that its fat is healthy contributes significantly to its weight loss features.
Avocados help in weight loss in different ways.
A randomized controlled trial was conducted by Naiman A Khan at the University of Illinois Urbana-Champaign to study the redistribution of belly fat by eating avocados.
The study included 105 overweight and obese adults.
The participants were given one meal per day for 12 weeks.
One group received a fresh avocado in their daily diet.
The other group ate a similar diet without avocado.
The participants’ glucose tolerance (a measure of metabolism) and abdominal fat were measured at the beginning and end of 12 weeks.
The following observations were made:
The researchers hope to conduct a follow-up trial where they plan to observe the changes in the gut and physical health in response to avocado consumption.
These results can aid nutritionists and other healthcare professionals in providing better dietary recommendations to reduce fat storage and lower diabetes risk.
The Best Ways To Consume Avocados To Reduce Fat
https://www.sciencedaily.com/releases/2021/09/210907110702.htm
https://www.medicalnewstoday.com/articles/270406
Whole grains (WG) offer a complete package of health benefits, unlike refined grains (RG), which are stripped off of valuable nutrients during the refining process. RG intake is associated with adverse health outcomes, including increased risk for cardiovascular diseases, type 2 diabetes, and obesity. Therefore, choosing WG over RG grains improves health in many ways. In this regard, a recent study has found that WG intake is tied to fewer heart disease risk factors.
WG comprises all the three parts of the grain, i.e. endosperm, germ, and bran.
They have been a part of the human diet for thousands of years.
They are a rich source of carbohydrates, multiple nutrients, and dietary fiber.
Some Of The Common Varieties Of WG Include:
Consumption of fiber-rich foods offers not only high nutrition but also prevents overeating.
WG and their products are more nutritious than RG and thus lower the risk of obesity.
The magnesium found in WG helps your body to break down carbs.
This helps increase insulin sensitivity, thereby, reducing the risk of type 2 diabetes.
According to research, WG bread and cereals are specifically linked to reduced risk of heart disease and stroke.
Framingham Offspring Cohort Study examined the association between WG and RG intake in 3121 participants.
The changes in waist circumference (WC), fasting HDL, triglycerides, glucose concentration, and blood pressure of the participants were recorded.
Replacing RG with WG among middle to older-aged adults can be an effective dietary change to reduce the risk of cardio-metabolic diseases.
https://medicalxpress.com/news/2021-09-grain-intake-tied-heart-disease.html
https://www.webmd.com/food-recipes/features/reap-the-benefits-of-whole-grains
https://pubmed.ncbi.nlm.nih.gov/27301975/
Cardiometabolic diseases include cardiovascular conditions such as heart attack, stroke, angina, and metabolic conditions like insulin resistance, type II diabetes, and non-alcoholic fatty liver disease. Over the years, low-fat diets have been embraced due to their health effects. But emerging evidence shows that low-carb diets may be just as effective. Recent research has suggested that low-carb diets have been shown to improve cardiometabolic risk profile.
In the last 50 years, the medical community has encouraged low-fat diets to avoid the effects of saturated fats on the heart. So low-fat and fat-free foods have been majorly circulating on the grocery shelves; however, many of these foods happen to be high in processed carbs.
However, recently many studies and healthcare professionals have been challenging this thought process. This has led to the emergence of the ketogenic diet.
A ketogenic diet is a very low carb, high-fat diet, restricting intake of sugar-sweetened beverages, baked goods, candies, and sweets.
Some versions may also limit healthy carb sources, such as grains, starchy vegetables, high-carb fruits, pasta, and legumes.
The diet is high in protein, fat, and healthy vegetables. They may increase good cholesterol levels and decrease blood pressure and triglyceride levels.
Other than helping with weight loss, low-carb diets increase good cholesterol levels, reduce blood sugar levels, lower triglyceride levels, and keep your metabolism in control.
Please note: Some harmful effects like fatigue, kidney stones, headache, loss in muscle tissue have been reported with low-carb diets. Consult a qualified nutritionist before making any significant dietary changes.
The Boston Children's Hospital led a large clinical trial to examine the effects of a low-carb diet on cardiometabolic disease risk.
The study included 164 adults who were overweight or obese. The participants had already lost 10-14 percent of their body weight by undergoing a reduced-calorie diet.
The participants were randomly assigned one of these three diets:
The participants received their customized meals, thus ensuring that all of them rigidly followed the protocol.
In all the prepared meals, saturated fats comprised 35% of the total fat present. In the low-carb meal, saturated fat contributed to 21% of the calories, and in the high-carb meal, it contributed to 7% of the calories.
Compared to the lower-fat higher-carb diets, the low-carb diet had the following benefits:
Though this study was done on adults, the researchers say that low-carb diets may benefit children too. In fact, pediatric cardiologists are also starting to embrace low-carb diets.
Diet is as important to mental health as it is to physical health. Previous studies have reported that the "traditional" dietary pattern, loaded with vegetable oil, meat, salt, and organ meat, is associated with increased odds of anxiety and depression in women. A recent study by researchers at the Ruhr-University Bochum and University of Duisburg, Germany, has reported higher depression scores among vegetarians than non-vegetarians.
Depression is a common yet serious mental condition that negatively impacts how you feel, the way you think and act. Depression occurs as a result of a combination of social, psychological, and biological factors.
Some of the common symptoms of depression are :
Depression susceptibility is related to diet both directly and indirectly. Unhealthy eating patterns can cause mood swings. When you stick to a healthy diet, you are setting yourself up for fewer mood fluctuations.
In particular, sugar is considered a major culprit. When consumed in higher quantities, it causes a temporary spike in 'feel-good' hormones like dopamine, which is not good for your health. In addition, the fleeting sugar rush followed by a crash is terrible for your mood.
A German research team conducted a meta-analysis on depression and vegetarian diet.
Meta-analysis refers to a procedure where the information collected from different experiments (with the same objective) is put together and studied. This combines the results of multiple studies to form a conclusion.
The analysis included data from 49,889 participants, of which 8,057 were vegetarians, and 41,832 were non-vegetarians. The large sample size makes this a robust study.
The researchers observed a higher depression score among vegetarians when compared to the non-vegetarians. But there was no causal relationship observed between them. That is, there was no proof that a vegetarian diet directly causes depressive moods. Depression didn't seem to increase a person's chance of adopting a vegetarian diet either.
This study, however, showed that it might be more likely that people switch to a vegetarian diet after developing mental health issues. The researchers cite three possible reasons for this:
Vitamin E is a group of 8 different nutrient compounds: 4 types of tocopherols and four types of tocotrienols. Both tocopherols and tocotrienols are types of vitamin E nutrients.
Image: Forms of Vitamin E
The most predominant form of vitamin E in the body is α-tocopherol. It comprises over 90% of the vitamin E found in the body. This form was first isolated from wheat germ oil. Interestingly, the “tokos” in α-tocopherol stands for “childbirth.”
Animal studies have revealed that a deficiency of α-tocopherol increases the risk of infertility. That’s why this nutrient is also known as anti-infertility vitamin or anti-sterility factor X.
Vitamin E is an essential nutrient, which means we need to obtain this nutrient through food sources.
Some foods rich in vitamin E are almonds, sunflower seeds, avocados, peanut butter, pine nuts, rainbow trout, and pumpkin.
Vitamin E is a potent antioxidant and protects our body from the damaging effects of free radicals.
Free radicals are unstable molecules that are harmful to the healthy cells in our bodies.
Vitamin E also has anti-aging properties.
Once vitamin E enters the body, it is absorbed by the intestines and stored in the adipose tissues, commonly known as body fat. On-demand, the adipose tissues are broken down to release vitamin E.
Here, it is important to know that the liver only acts on α-tocopherol and converts it into a form that is usable by the cells in the body. All other types of vitamin E are excreted out.
A healthy adult woman requires about 8 mg of vitamin E per day. In men and pregnant women, the requirement increases to 10 mg per day.
Vitamin E deficiency can result in a weakened immune system, muscle damage, vision loss, and nervous system-related disorders.
Many conditions like cystic fibrosis, short bowel syndrome, and chronic pancreatitis prevent effective absorption of fats, including the fat-soluble vitamin E. So, they can increase your risk for vitamin E deficiency.
Genetics is another important factor that contributes to vitamin E deficiency.
The TTPA gene is crucial for regulating vitamin E levels in the body. It contains instructions for the production of α-tocopherol transfer protein. This protein is responsible for the distribution of vitamin E obtained from the diet to all the cells and tissues of the body.
Any changes in this gene can affect the amount of the protein produced, and hence the vitamin E levels. People who have these changes are at a higher risk of vitamin E deficiency.
A simple genetic test can reveal your genetic status of vitamin E deficiency.
Most genetic tests provide your DNA information in the form of a text file called the raw DNA data. This data may seem like Greek and Latin to you.
At Xcode Life, can help you interpret this data. Upload your raw data and order a nutrition report.
Xcode Life then analyzes your raw data in detail to provide you with comprehensive nutrition analysis, including information on your vitamin E requirements.
Also Check Out: Gene Nutrition Report Walkthrough!
Folate (Vitamin B9) is also known as folic acid or folate. "Folic" is derived from the word "folium," meaning leaves, as in green leafy vegetables. Needless to say, leaves are one of the richest sources of vitamin B9.
The vitamin B9 we eat is absorbed in the jejunum region of the small intestine after going through minor structural changes.
In most cases, dietary sources of vitamin B9 are sufficient to meet this nutrient's requirement. Other than the leafy greens, some foods rich in vitamin B9 are beans, whole grains, seafood, peanuts, and sunflower seeds.
The Recommended Dietary Allowance or RDA for folate is 400 mcg/day for healthy adults. The RDA for lactating and pregnant women are 500 and 600 mcg/day, respectively.
Folate deficiency in pregnant women is one of the leading causes of neural tube defects, a birth abnormality, in babies.
Symptoms of vitamin B9 deficiency include extreme tiredness, pale skin, headaches, and heart palpitations.
Vitamin B9 needs to be converted into a form called tetrahydrofolate or THF to be effectively used by the body. The conversion of folic acid to THF is carried out by an enzyme called THF reductase.
This conversion is a very crucial step in the MTHFR cycle.
THF plays a very important role in converting a harmful amino acid called homocysteine to a safe and useful amino acid called methionine.
Image: Folate Cycle
The MTHFR gene is a well-known gene associated with folate deficiency. This gene helps the conversion of inactive vitamin B9 such as folate, or folic acid, to active B9, the THF.
30-60% of people have a change in this gene that ultimately leads to low vitamin B9 levels in the body.
Other genes like MTYL1 also influence your vitamin B9 levels.
Fortunately, vitamin B9 deficiency comes with a simple fix - increasing your dietary folate intake. In some cases, folate supplements may be advised.
A simple genetic test can reveal your genetic status of vitamin B9 deficiency.
Most genetic tests provide your DNA information in the form of a text file called the raw DNA data. This data may seem like Greek and Latin to you.
At Xcode Life, can help you interpret this data. Upload your raw data and order a nutrition report. Xcode Life then analyzes your raw data in detail to provide you with comprehensive nutrition analysis, including information on your vitamin B9 requirements.
Vitamin A is important for the overall development and maintenance of the body. Our body does not produce vitamin A on its own. It needs to be supplemented through diet; that's why it's called an essential vitamin.
The retina is the film screen, located at the very back of the eye. It contains two important cells that process the light entering our eyes.
The rod cells help us see in low light, while the cone cells help our color vision. The rod cells contain an important protein called rhodopsin, which moderates low light vision. A form of vitamin A called the retinal helps activate rhodopsin.
This is why a severe deficiency of vitamin A can cause night blindness.
Vitamin A is also crucial for maintaining skin integrity and forming new skin cells. Since vitamin A is an excellent antioxidant, including it in your diet every day can lower your risk for heart attack.
We all know that carrots are a good source of vitamin A. They are a rich source of a molecule called beta-carotene. Beta-carotene is a provitamin A. Provitamins are substances that are converted into active vitamins in the body.
Beta-carotene is what is responsible for the bright orange color of the carrot. All plants provide vitamin A in the form of beta-carotene, among other forms.
Vitamin A is present as retinol, a form of active vitamin A, in animal food sources. Now, the beta-carotene from plant sources must be converted to active vitamin A for it to be useful to the body.
Let’s see how that happens.
The structure of beta carotene resembles that of a dumbbell - two ring-like structures joined by a chain. This chain is cut in a particular way to give rise to two molecules of retinol, or active vitamin A. This cleavage happens in the liver.
Image: Cleavage of beta-carotene to retinol
Vitamin A in the body can be converted or interconverted into different formats. The retinol and retinal forms are interchangeable, while there’s only a one-way conversion from retinal to retinoic acid.
Image: Different forms of active vitamin A
The retinal form of vitamin A is absorbed by the intestinal villi along with fats. From there, it is transported to and stored in the liver. Whenever there's a requirement for vitamin A, retinal is released by the liver. It then binds to the specific retinol-binding protein, which serves as a carrier to transport it to various locations of the body.
The cleavage or the cutting of beta-carotene to form retinol is carried out by an enzyme called Beta Carotene Oxygenase or Monooxygenase. This enzyme is produced by the gene called BCMO1 or BCO1.
Every person has two copies of the BCMO1 gene. But, about 45 percent of the population carries at least one change or variation in the gene that reduces the enzyme activity. This results in a significantly impaired ability to convert beta-carotene into retinal.
Depending on which combination of variants someone has, beta-carotene conversion can be nearly 70 percent lower than its normal efficiency.
Vitamin A deficiency has serious health implications.
Knowing your BCMO1 gene status can help you gauge your genetic risk for vitamin A deficiency. This can be done through a genetic test.
Most genetic tests provide your DNA information in the form of a text file called the raw DNA data. This data may seem like Greek and Latin to you.
At Xcode Life, can help you interpret this data. All you have to do is upload your raw data and order a nutrition report. Xcode Life then analyzes your raw data in detail to provide you with comprehensive nutrition analysis, including information on your vitamin A requirements.
