Reports
Health
Wellness
To read your Promethease review report you will need to ensure that you are accustomed to some of the terms that are found in their report like magnitude and repute. It is difficult to sift through thousands of pages in your report. Once you organize the pages as per your unique needs it becomes a relatively easy task to.
Promethease allows users to analyze their genetic data and compare personal genomics results against the SNPedia database. SNPedia is a wiki-styled genetic data repository.
Though popular, it is often criticized for being too technical and difficult to read. You can take a look at Promethease's sample report here.
Promethease is a do-it-yourself (DIY) tool for health genetic information from 23andMe raw data. It is a third party online tool that provides consumers information about their genetic variants and how they affect your health. After 23andme FDA ban on providing health reports, people started using tools like Promethease to get more information from their DNA raw data, other than just locating distant cousins. Though Promethease is a good tool built on a repository like SNPedia there are a number of factors to consider before you decide to upload your raw data to Promethease. Promethease is popular as a low-cost offering, but it’s also very technical and disease-centric. There are many alternatives to Promethease to analyze your 23andme raw data, that are easy to follow and understand. Below we present a feature by feature comparison between Promethease and Xcode Life Sciences.
Promethease was one of the early companies to offer DNA raw data analysis based on 23andMe, Ancestry DNA raw data.
A DNA raw data is a file that contains information about your genetic variants. These are called single nucleotide polymorphisms or SNPs and account for the differences in different traits like eye and hair color, height, and skin pigmentation across individuals. Your ancestry DNA raw data is a lab-generated text file. It is usually in the following two formats
The whole-genome files have a size of 80-90 GB (30x). All the top consumer genetic testing companies like 23andMe, AncestryDNA, Family Tree DNA enable their customers to [download their raw data](https://www.xcode.life/23andme-raw-data/download-23andme-dna-raw-data-third-3rd-party-tool) directly from the website or request a download.
The raw data from different ancestry DNA testing companies have different numbers of genetic markers depending on the microarray chip used. For example, the 23andMe raw data has around 650,000 SNPs in its v5 chip and AncestryDNA has around 700,000 SNPs.
When the FDA (briefly) banned 23andMe from providing health reports, Promethease was the only alternative and gained popularity because of its free service and low turnaround time.
Promethease is criticized for its user-unfriendly interface based on several user reviews. Some well-known competitors for this tool are Xcode Life, Codegen, Interpretive, Nutrahacker, and GEDMatch.
MyHeritage offered Promethease free of charge through the end of 2019 and continues to maintain SNPedia as a free resource for academic and non-profit users. The raw data of Non-European users has been shifted to MyHeritage into new accounts created for them. However, users retain ownership of their DNA raw data file and are free to delete it from MyHeritage's server.
Promethease takes approximately 20 minutes to generate health report from your raw data.
Promethease offers health reports based on 23andme, Ancestry DNA, Family Tree DNA, and other ancestry raw DNA data providers for a price of $12. Once you upload your DNA raw data, you will get your report in about 20 minutes.
Earlier, the reports were offered free or at a discount. So many users tried them due to the free/low-cost aspect and the high accuracy that the vastness of their content seemed to imply. However, the reports left individuals more confused than clear.
So if you have a Promethease report that you still cannot make heads or tails of, read on.
Promethease is a wiki-style collection of peer-curated genetic information in contrast to expert-curated reports like Xcode Life's reports. You can search genes/variants/diseases as you would on Wikipedia. The results are pages of information. This is great for people who have degrees in genetics and want to learn about all the diseases they can get.
However, if you want organized information that you can use in specific aspects of your daily life, such as Nutrition, Fitness, or Ancestry, to enhance your health and wellbeing, then you may not find the Promethease reports all that helpful. Promethease's emphasis seems to be largely on the different diseases that one can get.
Once you transfer your DNA raw data to Promethease, you will get a lengthy report. Though it may look like a massive report on the face of it, the amount of potentially useful information in the Promethease report is quite limited. Understanding and interpreting this report is a challenge.
Let's break it down:
The total number of entries in a typical Promethease report = Approximately 25,000
The "Summary" component of the Promethease report is where the information pertaining to your genotype is given. This column, for the vast majority of the entries, has information that will not be useful to most people. It contains many terms that carry no useful information for the regular user.
The number of entries remaining after removing empty entries (no annotation) and terms such as "common in ClinVar," "common on affy axiom," "none," and "complete genomics,"," is 1482.
On further removal of more such terms (normal, common, etc.), you will be left with ~800 entries.
> So, only about 800 entries out of 25,000 (~ 3%) are potentially informative entries in the Promethease report.
A lot of people think that getting the additional health report from 23andme will help them get better results. That is not true! Whether you order the Health report or the Ancestry report from 23andme, the raw data you receive is the same!
This third party DNA raw data analysis tool, is a literature retrieval system from SNPedia which is a Wikipedia style repository where people can add information. It is not a systematic risk assessment tool. For example, for Alzheimer’s disease (AD), you will not get a summary of your risk for developing AD. Instead, you will get a list of scientific literature that is available that indicates or does not indicate your risk for AD for a specific variant. For a particular disease or trait, if there are 15 articles that report on that particular DNA location, based on your SNPs 2 information sources might indicate an increased risk, 5 might be neutral, and 3 towards the lower side. Promethease does not summarise these for you. Rather it returns all these 10 articles and makes you do the interpreting yourself. This is the major reason why many individuals are alarmed and confused by their Promethease report.
The vast majority of human traits are polygenic, meaning, several genes contribute towards that trait. Even if one carries a disease variant, they may also be carrying variants that reduce the risk of disease. Even in the case of Monogenic conditions- where one single gene leads to disease, scientist have found individuals carrying these deadly mutations but apparently healthy with no sign of disease. In a majority of the cases, the genetic risk is a small contributor towards the overall risk of that disease/condition. So, one must consider all of these things in interpreting their report.
Promethease only relies on SNPedia. Several other notable and high scientific authority tools are left out from the Promethease reports.
It is impractical to sift through the dump of information that you get (anticipate about 48,000 rows of data). The tool is equipped with different kinds of filters aimed at assisting you. Some of them are gene names, haplogroup names, diseases, drug names (referred to as “medicines”), medical conditions, names of genes and race.
Magnitude is a subjective parameter that measures the interest level of the information. Here is the table partially resourced from the SNPedia website that describes what each numerical value indicates.
| A-C | D-F | G-H | I-N | O-Z |
|---|---|---|---|---|
| 3 beta-Hydroxysteroid dehydrogenase deficiency | Deafness, autosomal recessive 77 | Glucose-6-phosphate dehydrogenase (G6PD) deficiency | Inclusion Body Myopathy 2 | Ornithine aminotransferase deficiency |
| 3-Phosphoglycerate Dehydrogenase Deficiency | Deficiency of acetyl-CoA acetyltransferase | Galactosylceramide beta-galactosidase deficiency | Infantile Sialic acid storage disease; Salla disease | Ornithine carbamoyltransferase deficiency |
| Abetalipoproteinaemia | Deficiency of alpha-mannosidase | Gaucher disease | Isovaleryl-CoA dehydrogenase deficiency | Partial adenosine deaminase deficiency |
| Achondrogenesis, type IB; Multiple epiphyseal dysplasia type 4; Atelosteogenesis type II; Diastrophic dysplasia | Deficiency of galactokinase | Gitelman Syndrome | Joubert Syndrome 2 / Meckel Syndrome 2 | Pendred syndrome |
| Achromatopsia | Deficiency of guanidinoacetate methyltransferase | Glutaric acidemia type 1 | Joubert syndrome 28 | Peroxisomal acyl-CoA oxidase deficiency |
| Acute infantile liver failure due to synthesis defect of mtDNA-encoded proteins | Deficiency of hydroxymethylglutaryl-CoA lyase | Glutaric acidemia type 2A | Joubert syndrome 5 | Phenylketonuria |
| Acyl-CoA dehydrogenase family, member 9 deficiency | Deficiency of steroid 11-beta-monooxygenase | Glutaric acidemia type 2C | Joubert syndrome 7 | Phosphoribosylpyrophosphate synthetase superactivity |
| Adrenoleukodystrophy | Deficiency of UDPglucose-hexose-1-phosphate uridylyltransferase | Glycine encephalopathy (AMT-related) | Junctional epidermolysis bullosa (LAMA3 related) | Polyglandular autoimmune syndrome, type 1 |
| Adult polyglucosan body neuropathy | Dihydrolipoamide dehydrogenase deficiency | Glycine encephalopathy (GLDC-related) | Junctional epidermolysis bullosa (LAMB3 related) | Polymicrogyria |
| Aicardi Goutieres syndrome 5 | Duchenne muscular dystrophy | Glycogen storage disease due to glucose-6-phosphatase deficiency type IA | Junctional epidermolysis bullosa (LAMC2 related) | Pontocerebellar hypoplasia type 1A |
| Alpha thalassemia-X-linked intellectual disability syndrome | Dyskeratosis congenita, autosomal recessive, 5 | Glycogen storage disease type 1B | Juvenile retinoschisis | Pontocerebellar hypoplasia type 2D |
| Alport Syndrome (COL4A3-related) | Dystrophic epidermolysis bullosa | Glycogen storage disease type 2 | Lamellar Ichthyosis, Type 1 | Pontocerebellar hypoplasia type 6 |
| Alport Syndrome (COL4A4-related) | Ehlers-Danlos syndrome dermatosparaxis type | Glycogen storage disease type 3 | Laminin alpha 2-related dystrophy | Primary carnitine deficiency |
| Alport Syndrome, X-linked (COL4A5-related) | Ellis-van Creveld syndrome (EVC-related) | Glycogen storage disease type 4 | Laryngo-onycho-cutaneous syndrome | Primary Ciliary Dyskinesia (DNAH11-related) |
| Alstrom syndrome | Ellis-van Creveld syndrome (EVC2-related) | Glycogen storage disease type 5 | Leber congenital amaurosis 10 | Primary Ciliary Dyskinesia (DNAH5-related) |
| Anauxetic dysplasia; Cartilage-hair hypoplasia; Metaphyseal dysplasia without hypotrichosis | Emery-Dreifuss muscular dystrophy, X-linked | Glycogen storage disease type 7 | Leber congenital amaurosis 13 | Primary Ciliary Dyskinesia (DNAI1-related) |
| Andermann Syndrome | Enhanced s-cone syndrome | GRACILE syndrome/ BCS1L-related disorders | Leber congenital amaurosis 2 | Primary Ciliary Dyskinesia (DNAI2-related) |
| Arginase deficiency | Ethylmalonic encephalopathy | Hemochromatosis type 2A | Leber congenital amaurosis 5 | Primary hyperoxaluria type I |
| Argininosuccinate lyase deficiency | Fabry disease | Hemochromatosis type 3 | Leber congenital amaurosis 8 | Primary hyperoxaluria type II |
| Aromatase deficiency | Familial dysautonomia | Hereditary acrodermatitis enteropathica | Leigh syndrome, French Canadian type | Primary hyperoxaluria type III |
| Arthrogryposis, mental retardation, and seizures | Familial hypercholesterolemia (LDLR related) | Hereditary factor IX deficiency disease | Lethal congenital contracture syndrome | Progressive familial intrahepatic cholestasis 2 |
| Arts syndrome | Familial hypercholesterolemia (LDLRAP1 related) | Hereditary fructosuria | Leukoencephalopathy with vanishing white matter | Propionic acidemia (PCCA related) |
| Asparagine synthetase deficiency | Familial Hyperinsulinism, ABCC8-Related | Hereditary insensitivity to pain with anhidrosis | Limb-girdle muscular dystrophy type 2A | Propionic acidemia (PCCB related) |
| Aspartylglucosaminuria | Familial isolated deficiency of vitamin E | Hereditary spastic paraplegia 15 | Limb-girdle muscular dystrophy type 2B | Pyknodysostosis |
| Ataxia-telangiectasia syndrome | Fanconi anemia type A | Hermansky-Pudlak syndrome type 1 | Limb-girdle muscular dystrophy type 2C | Pyruvate carboxylase deficiency |
| Autosomal recessive osteopetrosis 1 | Fanconi anemia type C | Hermansky-Pudlak syndrome type 3 | Limb-girdle muscular dystrophy type 2D | Pyruvate dehydrogenase E1-alpha deficiency |
| Autosomal recessive polycystic kidney disease | Fanconi anemia type G | Holocarboxylase synthetase deficiency | Limb-girdle muscular dystrophy type 2E | Pyruvate dehydrogenase E1-beta deficiency |
| Bardet-Biedl syndrome 1 | Fetal akinesia deformation sequence | Homocystinuria (CBS related) | Limb-girdle muscular dystrophy type 2F | Renal tubular acidosis with progressive nerve deafness |
| Bardet-Biedl syndrome 10 | Finnish congenital nephrotic syndrome | Homocystinuria due to MTHFR deficiency | Long-chain 3-hydroxyacyl-CoA dehydrogenase deficiency | Retinitis pigmentosa 20 |
| Bardet-Biedl syndrome 12 | Fragile X syndrome | Homocystinuria, cobalamin E type | Lysinuric protein intolerance | Retinitis pigmentosa 25 |
| Bardet-Biedl syndrome 13 | Fukuyama congenital muscular dystrophy | Hydrolethalus syndrome | Lysosomal acid lipase deficiency | Retinitis pigmentosa 26 |
| Bardet-Biedl syndrome 14 | Fumarase deficiency | Hyperlipoproteinemia, type I | Maple syrup urine disease type 1A | Retinitis pigmentosa 28 |
| Bardet-Biedl syndrome 2 | Hyperornithinemia-hyperammonemia-homocitrullinuria syndrome | Maple syrup urine disease type 1B | Retinitis pigmentosa 59 | |
| Bare lymphocyte syndrome 2 | Hypohidrotic X-linked ectodermal dysplasia | Maple syrup urine disease type 2 | Retinitis pigmentosa 73 | |
| Bartter disease type 4a | Hypophosphatasia | Meckel-Gruber syndrome | Retinitis pigmentosa 76 | |
| BH4-deficient hyperphenylalaninemia A | Meckel syndrome type 1 | Rhizomelic chondrodysplasia punctata type 1/ Refsum disease (PEX7-related) | ||
| Bifunctional peroxisomal enzyme deficiency | Meckel syndrome type 4 | Rhizomelic chondrodysplasia punctata type 3 | ||
| Bloom syndrome | Meckel syndrome, type 5 | Roberts-SC phocomelia syndrome | ||
| Canavan Disease | Medium-chain acyl-coenzyme A dehydrogenase deficiency | Sandhoff disease | ||
| Carnitine palmitoyltransferase I deficiency | Megalencephalic leukoencephalopathy with subcortical cysts 1 | Schimke immuno-osseous dysplasia | ||
| Carnitine palmitoyltransferase II deficiency | Menkes kinky-hair syndrome | Segawa syndrome | ||
| Carpenter syndrome 1 | Metachromatic leukodystrophy (ARSA related) | Senior-Loken syndrome 6 | ||
| Cerebrotendinous xanthomatosis | Metachromatic leukodystrophy due to | Severe combined immunodeficiency (RAG2-related) | ||
| Charcot-Marie-Tooth disease type 4D | saposin-b deficiency | Severe combined immunodeficiency due to ADA deficiency | ||
| Charcot-Marie-Tooth disease, X-linked | Methylmalonic acidemia (MMAA related) | Severe combined immunodeficiency due to DCLRE1C deficiency | ||
| Charcot-Marie-Tooth Neuropathy X | Methylmalonic acidemia (MMAB related) | Severe combined immunodeficiency, X-linked | ||
| Charlevoix-Saguenay spastic ataxia | Methylmalonic Aciduria and Homocystinuria, Type cblC | Severe congenital neutropenia 3 | ||
| Choreoacanthocytosis | Methylmalonic Aciduria and Homocystinuria, Type cblD | Severe congenital neutropenia 5, autosomal recessive | ||
| Choroideremia | Microcephaly, postnatal progressive, with seizures and brain atrophy | Severe X-linked myotubular myopathy | ||
| Chronic granulomatous disease (CYBA related) | Microphthalmia /clinical anophthalmia (VSX2-related) | Sjögren-Larsson syndrome | ||
| Chronic granulomatous disease, X-linked | Mitochondrial complex I deficiency/ Leigh syndrome (NDUFAF5-related) | Smith-Lemli-Opitz syndrome | ||
| Citrin deficiency | Mitochondrial complex I deficiency/ Leigh syndrome (NDUFAF6-related) | Spastic paraplegia 49, autosomal recessive | ||
| Citrullinemia type I | Mitochondrial DNA depletion syndrome 1 (MNGIE type) | Spinal muscular atrophy | ||
| Cockayne syndrome type A | MPV17-related mitochondrial DNA maintenance defect | Spondylocostal dysostosis | ||
| Cockayne syndrome type B | Mucolipidosis type II | Steel syndrome | ||
| Cohen syndrome | Mucolipidosis type III gamma | Stuve-Wiedemann Syndrome | ||
| Combined malonic and methylmalonic aciduria | Mucolipidosis type IV | Tay-Sachs disease | ||
| Combined oxidative phosphorylation deficiency 1 | Mucopolysaccharidosis type I (Hurler syndrome) | Tooth agenesis, selective, 4; Odonto-onycho-dermal dysplasia; Schopf-Schulz-Passarge syndrome | ||
| Combined oxidative phosphorylation deficiency 3 | Mucopolysaccharidosis type II (Hurler syndrome) | Tyrosinemia type I | ||
| Combined Pituitary Hormone Deficiency-2 | Mucopolysaccharidosis type IIIA | Tyrosinemia type II | ||
| Combined Pituitary Hormone Deficiency-3 | Mucopolysaccharidosis type IIIB (Sanfilippo B) | Usher syndrome type 1B | ||
| Congenital adrenal hyperplasia due to 17-alpha-hydroxylase deficiency | Mucopolysaccharidosis type IIIC | Usher syndrome type 1C | ||
| Congenital adrenal hyperplasia due to 21-hydroxylase deficiency | Mucopolysaccharidosis type IIID | Usher syndrome type 1D | ||
| Congenital Adrenal Hyperplasia | Mucopolysaccharidosis type IVB (Morquio B syndrome)/ GM1 gangliosidosis | Usher syndrome type 1F | ||
| Congenital amegakaryocytic thrombocytopenia | Mucopolysaccharidosis type IX | Usher syndrome type 2A | ||
| Congenital bilateral aplasia of vas deferens from CFTR mutation | Mucopolysaccharidosis type VI | Usher syndrome type 3A | ||
| Congenital disorder of glycosylation type 1A | Multiple sulfatase deficiency | Very long-chain acyl-CoA dehydrogenase deficiency | ||
| Congenital disorder of glycosylation type 1B | Muscular dystrophy-dystroglycanopathy, FKTN-related | Walker-Warburg congenital muscular dystrophy | ||
| Congenital disorder of glycosylation type 1C | Myopathy, lactic acidosis, and sideroblastic anemia 1 | Werdnig-Hoffmann disease | ||
| Congenital hyperammonemia, type I | N-acetylglutamate Synthase Deficiency | Wilson disease | ||
| Congenital Hyperinsulinism | Nemaline myopathy 2 | Xeroderma pigmentosum group A | ||
| Congenital muscular dystrophy-dystroglycanopathy | Nephrogenic diabetes insipidus | Xeroderma pigmentosum, group C | ||
| Congenital myasthenic syndrome | Nephrotic syndrome, idiopathic, steroid-resistant | Zellweger spectrum disorder (PEX1-related) | ||
| Congenital myasthenic syndrome (CHRNE) | Neuronal ceroid lipofuscinosis 1 | Zellweger spectrum disorder (PEX10-related) | ||
| Corneal dystrophy-perceptive deafness syndrome | Neuronal ceroid lipofuscinosis 2 | Zellweger spectrum disorder (PEX12-related) | ||
| Corneal endothelial dystrophy | Neuronal ceroid lipofuscinosis 3 | Zellweger spectrum disorder (PEX2-related) | ||
| Corticosterone Methyloxidase Deficiency | Neuronal ceroid lipofuscinosis 5 | Zellweger spectrum disorder (PEX6-related) | ||
| Costeff Syndrome (3-Methylglutaconic Aciduria, Type 3) | Neuronal ceroid lipofuscinosis 6 | |||
| Creatine transporter deficiency | Neuronal ceroid lipofuscinosis 7 | |||
| Cystic fibrosis | Neuronal ceroid lipofuscinosis 8 | |||
| Cystinosis | Niemann-Pick disease type C1 | |||
| Niemann-Pick disease type C2 | ||||
| Niemann-Pick Disease, Types A/B | ||||
| Nijmegen breakage syndrome | ||||
| Nonsyndromic hearing loss and deafness |
Good repute: The variation has a positive influence on you.
Not set: There is no verified information on the nature of the variation.
Bad repute: The variation has a negative influence on you.
According to the SNPedia website, it is better to leave the repute as “Not Set” as all genotypes have a little of both the “good” and “bad” repute.
This option allows you to see only the SNPs present in ClinVar. ClinVar is a freely accessible public archive of information on genetic variations specifically with clinical implications. It is maintained by the National Institute of Health (NIH). When you filter using this option you will find all the medical conditions that have been formally labeled by ClinVar.
How is Xcode Life Report different from the Promethease report?
Xcode Life puts together expert-curated references of variant annotations from several large databases and leading scientific journals.
The information is then organized systematically into topical reports such as Nutrition, Health, Fitness, Skin, Allergy, Ancestry, etc.
Each report is further organized into traits, which provide actionable insights into your genetic type along with specific recommendations. Xcode reports are easily readable, understandable, and implementable.
The core philosophy of Xcode reports, in contrast to Promethease's voluminous coverage of diseases, is to empower the user with actionable genetic information that they can use to enhance health and wellbeing. Each report is reasonably priced at around $20; additionally, there are package discounts if the user buys multiple reports together.
If you are not looking for anything specific and just want to satisfy your curiosity, then Promethease may be the way to go. Even then, it is very likely that you will be left scratching your head with a vast load of information that you can't make sense of. But, if you want specific, organized, and actionable insights from your genetic data about your health and wellbeing, then you certainly must try the Xcode Health reports.
| Drug (A-C) | Gene | Guidelines | Drug (D-N) | Gene | Guidelines | Drug (O-Z) | Gene | Guidelines |
|---|---|---|---|---|---|---|---|---|
| (R)-methadone;(S)-EDDP;methadone | UGT2B7 | PGX | Dabigatran | CES1 | PGX | o-desmethyltramadol;tramadol | RFPL4B | PGX |
| 3,4-methylenedioxymethamphetamine | SLC6A2 | PGX | daclatasvir;peginterferon alfa-2a;peginterferon alfa-2b;ribavirin | IFNL3;IFNL4 | PGX | olanzapine | ABCB1 | PGX |
| abacavir | HLA-B | CPIC | dalcetrapib | ADCY9 | PGX | olanzapine | BDNF | PGX |
| abacavir | HLA-B | DPWG | dapsone | HLA-DRB1 | PGX | olanzapine | GIPR | PGX |
| abacavir | HLA-B | FDA | daptomycin | ABCB1 | PGX | olanzapine;perphenazine | RGS4 | PGX |
| abacavir | LST1;LTA;LTB | PGX | darifenacin | CYP2D6 | FDA | olanzapine;risperidone | DRD2 | PGX |
| abiraterone;prednisolone | DSE;TSPYL1 | PGX | dasatinib;imatinib | ABCG2 | PGX | olanzapine;risperidone | HTR2A | PGX |
| ABT-751 | UGT1A4 | PGX | dasatinib;imatinib;nilotinib | ABCG2 | PGX | omeprazole | CYP2C19 | CPIC |
| acamprosate | GRIN2B | PGX | daunorubicin | NOS3 | PGX | omeprazole | CYP2C19 | DPWG |
| Ace Inhibitors;Plain | XPNPEP2 | PGX | debrisoquine | CYP2D6 | PGX | omeprazole | CYP2C19 | FDA |
| Ace Inhibitors;Plain;Angiotensin II Antagonists;Beta Blocking Agents;digoxin;diuretics;spironolactone | ADRB2 | PGX | deferasirox | ABCG2 | PGX | omeprazole | ABCB1 | PGX |
| Ace Inhibitors;Plain;Angiotensin II Antagonists;Beta Blocking Agents;digoxin;diuretics;spironolactone | NOS3 | PGX | deferasirox | UGT1A3 | PGX | ondansetron | CYP2D6 | CPIC |
| acenocoumarol | CYP4F2 | PGX | deferiprone | UGT1A6 | PGX | ondansetron | ABCB1 | PGX |
| acenocoumarol | VKORC1 | PGX | deleobuvir;faldaprevir | IFNL3;IFNL4 | PGX | Opioid anesthetics;Other general anesthetics;volatile anesthetics | OPRM1 | PGX |
| acenocoumarol;phenprocoumon | VKORC1 | PGX | desflurane | CACNA1S | PGX | opioids | OPRM1 | PGX |
| acenocoumarol;warfarin | APOE | PGX | desflurane;enflurane;halothane;isoflurane;methoxyflurane;sevoflurane;succinylcholine | RYR1 | PGX | Opium alkaloids and derivatives | CHRNA3 | PGX |
| acetaldehyde | ALDH2 | PGX | desipramine | CYP2D6 | CPIC | Opium alkaloids and derivatives | CHRNA5 | PGX |
| acetaldehyde | PGM1 | PGX | desipramine | CYP2D6 | FDA | oseltamivir | ABCB1 | PGX |
| acetaminophen | TRPV1 | PGX | desipramine | MC1R | PGX | oxaliplatin | ABCB1 | PGX |
| acetaminophen | UGT1A | PGX | desloratadine | HRH1 | PGX | oxazepam | UGT2B15 | PGX |
| acetaminophen;aspirin;diclofenac;propionic acid derivatives;Pyrazolones | PLA2G4A | PGX | desloratadine;mizolastine | FCER1A | PGX | oxcarbazepine | HLA-B | CPIC |
| acetaminophen;ibuprofen;loxoprofen;salicylamide | PGX | deutetrabenazine | CYP2D6 | FDA | oxcarbazepine | HLA-B | FDA | |
| acetaminophen;tramadol | OPRM1 | PGX | dexamethasone | SERPINE1 | PGX | oxcarbazepine | ABCB1 | PGX |
| acetylcholine | ACE | PGX | dexamethasone;doxorubicin;vincristine | ABCB1 | PGX | oxycodone | COMT | PGX |
| acetylcysteine | TOLLIP | PGX | dexamethasone;lenalidomide | ABCB1 | PGX | paclitaxel | ABCB1 | PGX |
| adalimumab | ATG16L1 | PGX | dexlansoprazole | CYP2C19 | CPIC | paclitaxel;Platinum compounds | CASP7 | PGX |
| adalimumab | HFE | PGX | dexlansoprazole | CYP2C19 | FDA | paliperidone | MC4R | PGX |
| adalimumab;certolizumab pegol;etanercept;glucocorticoids;infliximab;methotrexate | KLRD1 | PGX | dexmedetomidine | ADRA2A | PGX | pantoprazole | CYP2C19 | CPIC |
| adalimumab;etanercept;infliximab | FCGR3A | PGX | dextroamphetamine;methylphenidate | DRD1 | PGX | pantoprazole | CYP2C19 | DPWG |
| adalimumab;etanercept;infliximab;Tumor necrosis factor alpha (TNF-alpha) inhibitors | TRAF1 | PGX | diazepam | CYP2C19 | FDA | pantoprazole | CYP2C19 | FDA |
| adalimumab;infliximab | FCGR2A | PGX | dicloxacillin | ABCB1 | PGX | pantoprazole | ABCB1 | PGX |
| adefovir dipivoxil | SLC22A6 | PGX | didanosine | NT5C2 | PGX | paroxetine | CYP2D6 | CPIC |
| adefovir dipivoxil;cidofovir;tenofovir | SLC22A6 | PGX | difluorodeoxyuridine | CDA | PGX | paroxetine | CYP2D6 | DPWG |
| adrenergics, inhalants | PGX | digoxin | ABCB1 | PGX | paroxetine | CYP2D6 | FDA | |
| agomelatine | ABCB1 | PGX | Dihydropyridine derivatives | NOS1AP | PGX | paroxetine | CYP1A2 | PGX |
| alemtuzumab;chlorambucil;cyclophosphamide;fludarabine;prednisone;rituximab;vincristine | CXCL12 | PGX | diltiazem | PLCD3 | PGX | paroxetine | HTR1A | PGX |
| alendronate | VDR | PGX | dimethyl fumarate | GSTP1 | PGX | pazopanib | HLA-B | FDA |
| alfentanil | OPRM1 | PGX | Dipeptidyl peptidase 4 (DPP-4) inhibitors | CDKAL1 | PGX | pazopanib | UGT1A1 | FDA |
| Alkylating Agents | EGFR | PGX | direct acting antivirals | IFNL4 | PGX | peginterferon alfa-2a | IFNL3;IFNL4 | PGX |
| Alkylating Agents;cisplatin | ESR1 | PGX | disopyramide | KCNH2 | PGX | peginterferon alfa-2a;peginterferon alfa-2b | IFNL3;IFNL4 | PGX |
| allopurinol | HLA-B | CPIC | disulfiram | SLC6A3 | PGX | peginterferon alfa-2a;peginterferon alfa-2b;ribavirin | IFNL3 | PGX |
| allopurinol | HLA-B | FDA | diuretics | PDE4D | PGX | peginterferon alfa-2a;peginterferon alfa-2b;ribavirin | IFNL3;IFNL4 | PGX |
| allopurinol | ABCG2 | PGX | diuretics;hydrochlorothiazide | NEDD4L | PGX | peginterferon alfa-2a;peginterferon alfa-2b;ribavirin | IFNL4 | PGX |
| allopurinol | CYCSP5 | PGX | dobutamine | ADRB1 | PGX | peginterferon alfa-2a;peginterferon alfa-2b;ribavirin;simeprevir | IFNL4 | PGX |
| allopurinol;febuxostat | AOX1 | PGX | docetaxel | CYP3A4 | PGX | peginterferon alfa-2a;peginterferon alfa-2b;ribavirin;telaprevir | IFNL3 | PGX |
| allopurinol;febuxostat | UGT1A1;UGT1A10;UGT1A3;UGT1A4;UGT1A5;UGT1A6;UGT1A7;UGT1A8;UGT1A9 | PGX | docetaxel | SLCO1B3 | PGX | peginterferon alfa-2a;peginterferon alfa-2b;ribavirin;telaprevir | IFNL3;IFNL4 | PGX |
| alprazolam | CYP3A;CYP3A4 | PGX | docetaxel | VEGFA | PGX | peginterferon alfa-2a;ribavirin | SCARB1 | PGX |
| amantadine;Anticholinergics;Dopamine agonists;levodopa;selegiline | SLC22A1 | PGX | docetaxel;doxorubicin | AKR1C3 | PGX | peginterferon alfa-2a;ribavirin | VDR | PGX |
| amifampridine | NAT2 | FDA | docetaxel;epirubicin | MDM4 | PGX | peginterferon alfa-2b | HLA-DPA1 | PGX |
| amifampridine phosphate | NAT2 | FDA | docetaxel;FEC100;gemcitabine;trastuzumab;zoledronate | PPCDC | PGX | peginterferon alfa-2b;protease inhibitors;ribavirin | SLC28A2 | PGX |
| amiloride;spironolactone | CYP4A11 | PGX | docetaxel;gemcitabine;paclitaxel;Platinum compounds;vinorelbine | CASP7 | PGX | peginterferon alfa-2b;ribavirin | ITPA | PGX |
| aminoglycoside antibacterials | MT-RNR1 | PGX | docetaxel;thalidomide | CHST3 | PGX | pemetrexed | GGH | PGX |
| amiodarone | NOS1AP | PGX | dolasetron;granisetron | NOS1AP | PGX | pemetrexed | MTHFR | PGX |
| amiodarone;Antibiotics;antipsychotics;diuretics;quinidine;sotalol | KCNE1 | PGX | dolutegravir | UGT1A1 | FDA | perindopril | BDKRB1 | PGX |
| amisulpride | MC4R | PGX | dolutegravir | ABCG2 | PGX | perphenazine | CYP2D6 | FDA |
| amisulpride;antipsychotics;olanzapine;quetiapine;risperidone | HTR1A | PGX | donepezil | CYP2D6 | FDA | Pertussis vaccines | TLR4 | PGX |
| amisulpride;aripiprazole;clozapine;lithium;mirtazapine;olanzapine;paliperidone;quetiapine;risperidone;valproic acid | SH2B1 | PGX | donepezil | BCHE | PGX | phenazepam | CYP2C19 | PGX |
| amitriptyline | CYP2C19 | CPIC | donepezil | CHAT | PGX | phenobarbital | ABCB1 | PGX |
| amitriptyline | CYP2D6 | CPIC | donepezil;galantamine;rivastigmine | CHAT | PGX | phenobarbital;phenytoin | MTHFR | PGX |
| amitriptyline | CYP2D6 | DPWG | donepezil;galantamine;rivastigmine | CHRNA7 | PGX | phenprocoumon | VKORC1 | PGX |
| amitriptyline | CYP2D6 | FDA | doxepin | CYP2C19 | CPIC | phenytoin | CYP2C9 | DPWG |
| amitriptyline;antidepressants;citalopram;fluvoxamine;paroxetine;sertraline;venlafaxine | ABCB1 | PGX | doxepin | CYP2C19 | FDA | phenytoin | CFHR4 | PGX |
| amitriptyline;antidepressants;citalopram;paroxetine;venlafaxine | ABCB1 | PGX | doxepin | CYP2D6 | CPIC | phenytoin | SCN1A | PGX |
| amitriptyline;citalopram;fluoxetine;paroxetine;sertraline;venlafaxine | ABCB1 | PGX | doxepin | CYP2D6 | DPWG | photodynamic therapy | F13A1 | PGX |
| amitriptyline;citalopram;paroxetine;venlafaxine | ABCB1 | PGX | doxepin | CYP2D6 | FDA | pimozide | CYP2D6 | DPWG |
| amlodipine | CYP3A4 | PGX | doxorubicin | ABCC1 | PGX | pimozide | CYP2D6 | FDA |
| amlodipine;chlorthalidone | NPPA | PGX | doxorubicin | CBR3 | PGX | pioglitazone | ADIPOQ | PGX |
| amlodipine;chlorthalidone;lisinopril | ACE | PGX | doxorubicin;doxorubicinol | CBR1 | PGX | piroxicam | CYP2C9 | CPIC |
| amlodipine;chlorthalidone;lisinopril | AGT | PGX | doxorubicin;idarubicin | CYBA | PGX | piroxicam | CYP2C9 | FDA |
| amodiaquine | CYP2C8 | PGX | doxorubicin;methotrexate;prednisolone;vincristine | ABCB1 | PGX | pitavastatin | ABCC2 | PGX |
| amodiaquine;pyrimethamine;sulfadoxine | G6PD | PGX | dronabinol | CYP2C9 | FDA | pitrakinra | IL4R | PGX |
| amoxapine | CYP2D6 | FDA | Drug(s) | Gene | PGX | platinum | ERCC5 | PGX |
| amoxicillin;clavulanate | HLA-DQB1 | PGX | Drugs For Treatment Of Tuberculosis | NAT2 | PGX | Platinum compounds | GSTP1 | PGX |
| amphetamine | CYP2D6 | FDA | Drugs For Treatment Of Tuberculosis;isoniazid | ABCB1 | PGX | Platinum compounds | HSPA5 | PGX |
| amphetamine | DRD2 | PGX | Drugs For Treatment Of Tuberculosis;rifampin | RIPOR2 | PGX | Platinum compounds | XRCC1 | PGX |
| amphetamine;cannabinoids;cocaine;opioids | KAT2B | PGX | Drugs used in alcohol dependence | GATA4 | PGX | Platinum compounds;radiotherapy | PGX | |
| amprenavir | SLCO1B1 | PGX | Drugs Used In Diabetes | IRS1 | PGX | Platinum compounds;taxanes | ABCB1 | PGX |
| Analgesics | KCNJ6 | PGX | Drugs used in nicotine dependence;nicotine | CHRNA3 | PGX | pramipexole | DRD3 | PGX |
| Analgesics;Antiinflammatory agents, non-steroids;Ergot alkaloids;opioids;sumatriptan | COMT | PGX | Drugs used in nicotine dependence;nicotine;varenicline | CHRNA5 | PGX | prasugrel | CYP2C19 | PGX |
| Analgesics;Antiinflammatory agents, non-steroids;Ergot alkaloids;opioids;sumatriptan | OPRM1 | PGX | duloxetine | NCAM1 | PGX | pravastatin | KIF6 | PGX |
| anastrozole | CYP19A1 | PGX | duloxetine | ZNF385D | PGX | pravastatin | SLCO1B1 | PGX |
| anastrozole;exemestane | MAP4K4 | PGX | eculizumab | C5 | PGX | pravastatin;simvastatin | HMGCR | PGX |
| anastrozole;exemestane;letrozole | CYP19A1 | PGX | eculizumab | CR1 | PGX | prednisone;tacrolimus | ABCB1 | PGX |
| anastrozole;letrozole | TNFRSF11B | PGX | efavirenz | CYP2B6 | CPIC | pregabalin | SLC7A5 | PGX |
| anastrozole;letrozole | TNFSF11 | PGX | efavirenz | CYP2B6 | DPWG | prochlorperazine | ANKK1;DRD2 | PGX |
| angiotensin II | AGTR1 | PGX | efavirenz | CYP2B6 | FDA | propafenone | CYP2D6 | DPWG |
| anthracyclines and related substances | CBR3 | PGX | efavirenz | CYP2B6 | PGX | propafenone | CYP2D6 | FDA |
| anthracyclines and related substances | HAS3 | PGX | efavirenz;lopinavir;Nucleoside and nucleotide reverse transcriptase inhibitors;ritonavir | PGX | propofol | CYP2B6 | PGX | |
| anthracyclines and related substances;cyclophosphamide;doxorubicin;epirubicin;fluorouracil;methotrexate;oxaliplatin;Platinum compounds | NOS3 | PGX | efavirenz;nelfinavir | ABCB1 | PGX | propofol;remifentanil | ABCB1 | PGX |
| anthracyclines and related substances;taxanes | ABCB1 | PGX | egfr inhibitors | EGFR | PGX | propranolol | CYP2D6 | FDA |
| Antibiotics | LGALS3 | PGX | elagolix | SLCO1B1 | FDA | propranolol | ADRB2 | PGX |
| antidepressants | HTR2A | PGX | eliglustat | CYP2D6 | DPWG | protriptyline | CYP2D6 | FDA |
| antidepressants | SLC6A4 | PGX | eliglustat | CYP2D6 | FDA | quetiapine | COMT | PGX |
| antidepressants;antiepileptics;Beta Blocking Agents;calcium channel blockers | ACE | PGX | enalapril | PGX | quetiapine | MC4R | PGX | |
| antidepressants;antipsychotics;benzodiazepine derivatives;opioids;Sympathomimetics | OPRM1 | PGX | enalapril | ACE | PGX | quinapril | AGT | PGX |
| antidepressants;benzodiazepine derivatives;mirtazapine;Selective serotonin reuptake inhibitors | GAL | PGX | enalapril;imidapril;lisinopril | BDKRB2 | PGX | rabeprazole | CYP2C19 | FDA |
| antidepressants;citalopram | CREB1;METTL21A | PGX | endoxifen | PGX | radiotherapy | CDK1 | PGX | |
| antidepressants;citalopram;clomipramine;lithium;nefazodone;paroxetine;venlafaxine | FKBP5 | PGX | enflurane | CACNA1S | PGX | raloxifene | UGT1A8 | PGX |
| antidepressants;citalopram;escitalopram;fluoxetine;paroxetine | TPH1 | PGX | entacapone | COMT | PGX | raltegravir | UGT1A1 | FDA |
| antidepressants;citalopram;fluoxetine | SERPINE1 | PGX | Enzymes | A2M | PGX | ramipril | ACE | PGX |
| antidepressants;citalopram;fluvoxamine;paroxetine;sertraline;venlafaxine | ABCB1 | PGX | ephedrine;phenylephrine | ADRB2 | PGX | ranibizumab | NRP1 | PGX |
| antidepressants;citalopram;paroxetine | BDNF | PGX | epirubicin;fluorouracil;oxaliplatin | NQO1 | PGX | rasagiline | DRD2 | PGX |
| antidepressants;desipramine;fluoxetine | BDNF | PGX | erdafitinib | CYP2C9 | FDA | regadenoson | AMPD1 | PGX |
| antidepressants;fluvoxamine;paroxetine;Selective serotonin reuptake inhibitors;sertraline | HTR1A | PGX | erlotinib | EGFR | PGX | remifentanil | COMT | PGX |
| antidepressants;mirtazapine;venlafaxine | TPH2 | PGX | erythromycin | ABCC2 | PGX | remifentanil;sevoflurane | ABCB1 | PGX |
| antidepressants;Selective serotonin reuptake inhibitors | FKBP5 | PGX | escitalopram | CYP2C19 | CPIC | repaglinide | CYP2C8 | PGX |
| antidepressants;Selective serotonin reuptake inhibitors;venlafaxine | GRIA3 | PGX | escitalopram | CYP2C19 | DPWG | repaglinide | NEUROD1 | PGX |
| antiepileptics | ABCB1 | PGX | escitalopram | CYP2C19 | FDA | rhodamine 123 | ABCB1 | PGX |
| antiepileptics;carbamazepine | ABCB1 | PGX | escitalopram | BMP5 | PGX | rifampin | NR1I2 | PGX |
| antiepileptics;carbamazepine;lamotrigine;oxcarbazepine;phenytoin;topiramate | SCN2A | PGX | escitalopram | CYP1A2 | PGX | risperidone | CYP2D6 | DPWG |
| antiepileptics;carbamazepine;oxcarbazepine | ABCC2 | PGX | escitalopram;nortriptyline | BDNF | PGX | risperidone | CYP2D6 | FDA |
| antiepileptics;carbamazepine;phenobarbital;phenytoin;valproic acid | SCN2A | PGX | esomeprazole | CYP2C19 | FDA | risperidone | HTR2C | PGX |
| Antihypertensives | ATP2B1 | PGX | esomeprazole | STAT6 | PGX | risperidone | MC4R | PGX |
| Antihypertensives And Diuretics In Combination | NOS3 | PGX | estradiol | PGX | ritodrine | CACNA1C | PGX | |
| Antihypertensives;Antihypertensives And Diuretics In Combination;diuretics | NOS3 | PGX | etanercept | TNF | PGX | ritonavir | APOC3 | PGX |
| Antihypertensives;Beta Blocking Agents | GRK5 | PGX | ethambutol;isoniazid;pyrazinamide;rifampin | CYP2C9 | PGX | rituximab | FCGR3A | PGX |
| Antiinflammatory agents, non-steroids | CDKN2B-AS1 | PGX | ethanol | ADH1B | PGX | rivastigmine | BCHE | PGX |
| Antiinflammatory agents, non-steroids | PTGS1 | PGX | ethanol | OPRM1 | PGX | rocuronium | ABCB1 | PGX |
| Antiinflammatory agents, non-steroids;aspirin | PGX | etoposide | ABCB1 | PGX | rofecoxib | PTGS2 | PGX | |
| Antiinflammatory agents, non-steroids;celecoxib;diclofenac | CYP2C9 | PGX | etoposide | SLIT1 | PGX | rosiglitazone | PAX4 | PGX |
| antineoplastic agents | SERPINA3 | PGX | etravirine | CYP2C19 | PGX | rosuvastatin | SLCO1B1 | FDA |
| antineoplastic agents | TP53 | PGX | everolimus | PIK3R1 | PGX | rosuvastatin | ABCG2 | PGX |
| antipsychotics | BDNF | PGX | everolimus | RPTOR | PGX | rosuvastatin | SLCO1B1 | PGX |
| antipsychotics | GRM3 | PGX | exemestane | CYP19A1 | PGX | salbutamol | ADRB2 | PGX |
| antipsychotics;aripiprazole;bromperidol;chlorpromazine;clozapine;nemonapride;olanzapine;risperidone | DRD2 | PGX | exemestane;letrozole | CYP19A1 | PGX | salbutamol;selective beta-2-adrenoreceptor agonists | CRHR2 | PGX |
| antipsychotics;aripiprazole;risperidone | ANKK1 | PGX | exemestane;letrozole | ESR1 | PGX | salmeterol | ADRB2 | PGX |
| antipsychotics;chlorpromazine;fluphenazine;thioridazine;trifluoperazine | CYP1A2 | PGX | Farglitazar;glibenclamide | SCNN1B | PGX | salvianolic acid b | NOS3 | PGX |
| antipsychotics;clozapine;olanzapine | DRD2 | PGX | fenofibrate | APOA5 | PGX | selective beta-2-adrenoreceptor agonists | ARG1 | PGX |
| antipsychotics;olanzapine;perphenazine;quetiapine;ziprasidone | RGS4 | PGX | fenofibrate | CRP | PGX | Selective serotonin reuptake inhibitors | HTR7 | PGX |
| antipsychotics;risperidone | LEP | PGX | fentanyl | ABCB1 | PGX | sertraline | CYP2C19 | CPIC |
| antithymocyte globulin | FCGR3A | PGX | fentanyl | OPRM1 | PGX | sertraline | CYP2C19 | DPWG |
| Antithyroid Preparations | HLA-B | PGX | fentanyl;morphine;opioids;oxycodone | RHBDF2 | PGX | sertraline | GNB3;P3H3 | PGX |
| Antivirals for treatment of HIV infections, combinations;Drugs For Treatment Of Tuberculosis | ABCB1 | PGX | fentanyl;propofol;remifentanil;sevoflurane | ADRB2 | PGX | sevoflurane | CACNA1S | PGX |
| Antivirals for treatment of HIV infections, combinations;ritonavir | APOE | PGX | fesoterodine | CYP2D6 | FDA | sibutramine | GNB3 | PGX |
| apixaban | ABCG2 | PGX | fexofenadine | ABCB1 | PGX | sildenafil | GNB3 | PGX |
| aripiprazole | CYP2D6 | DPWG | fexofenadine | SLCO2B1 | PGX | silibinin | ABCB1 | PGX |
| aripiprazole | CYP2D6 | FDA | flecainide | CYP2D6 | DPWG | simvastatin | SLCO1B1 | FDA |
| aripiprazole | DRD2 | PGX | flecainide | SCN4A | PGX | simvastatin | HMGCR | PGX |
| aripiprazole lauroxil | CYP2D6 | FDA | flecainide;mexiletine | SCN4A | PGX | simvastatin | SLCO1B1 | PGX |
| aripiprazole;clozapine;haloperidol;olanzapine;quetiapine;risperidone | FAAH | PGX | flecanide | CYP2D6 | DPWG | simvastatin acid | SLCO1B1 | PGX |
| aripiprazole;risperidone | DRD2 | PGX | flibanserin | CYP2C19 | FDA | siponimod | CYP2C9 | FDA |
| artesunate;chlorproguanil;dapsone | G6PD | PGX | flucloxacillin | NR1I2 | PGX | sirolimus | ABCB1 | PGX |
| artesunate;primaquine;pyrimethamine;sulfadoxine | G6PD;IKBKG | PGX | fluindione | VKORC1 | PGX | sirolimus;temsirolimus | NR1I2 | PGX |
| asparaginase | NFATC2 | PGX | flunisolide | TBXT | PGX | sitagliptin;vildagliptin | GLP1R | PGX |
| asparaginase;cyclophosphamide;cytarabine;daunorubicin;dexamethasone;doxorubicin;leucovorin;mercaptopurine;methotrexate;pegaspargase;prednisone;thioguanine;vincristine | GATA3 | PGX | fluorouracil | DPYD | CPIC | SN-38 | UGT1A9 | PGX |
| asparaginase;cyclophosphamide;daunorubicin;prednisolone;vincristine | PNPLA3 | PGX | fluorouracil | DPYD | DPWG | sofosbuvir | IFNL3;IFNL4 | PGX |
| asparaginase;dexamethasone;methotrexate | PYGL | PGX | fluorouracil | DPYD | FDA | sofosbuvir;velpatasvir;voxilaprevir | IFNL3;IFNL4 | PGX |
| aspirin | PPARG | PGX | fluorouracil | DPYD | PGX | somatropin recombinant | COL1A1 | PGX |
| aspirin;Beta Blocking Agents;clopidogrel;hmg coa reductase inhibitors | NOS3 | PGX | fluorouracil;FOLFIRI;FOLFOX | TYMS | PGX | sorafenib | KDR | PGX |
| aspirin;clopidogrel | ITGA2 | PGX | fluorouracil;FOLFOX | TYMS | PGX | spironolactone | ACE | PGX |
| aspirin;clopidogrel;prasugrel | PEAR1 | PGX | fluorouracil;irinotecan;leucovorin | ABCG1 | PGX | stavudine | SAMHD1 | PGX |
| aspirin;prasugrel | NTRK1;PEAR1 | PGX | fluorouracil;irinotecan;leucovorin | ABCG2 | PGX | succinylcholine | CACNA1S | PGX |
| ataluren | CFTR | PGX | fluorouracil;leucovorin;oxaliplatin | MTHFR | PGX | succinylcholine | BCHE | FDA |
| atazanavir | UGT1A1 | CPIC | fluorouracil;oxaliplatin | PARD3B | PGX | succinylcholine;volatile anesthetics | RYR1 | PGX |
| atazanavir | UGT1A2 | CPIC | fluorouracil;Platinum compounds;radiotherapy | TMEM167A;XRCC4 | PGX | sufentanil | COMT | PGX |
| atazanavir | UGT1A3 | CPIC | fluorouracil;Platinum compounds;radiotherapy | XRCC4 | PGX | sufentanil | OPRM1 | PGX |
| atazanavir | NR1I2 | PGX | fluoxetine | SLC6A4 | PGX | sulfamethoxazole | GCLC | PGX |
| atazanavir | UGT1A1 | PGX | fluoxetine;sertraline | ACE | PGX | sulfasalazine | ABCG2 | PGX |
| atazanavir;lopinavir;ritonavir;tenofovir | ABCC2 | PGX | flurbiprofen | CYP2C9 | FDA | sulfinpyrazone | UGT1A9 | PGX |
| atazanavir;ritonavir | UGT1A7 | PGX | flurbiprofen | CYP2C9 | PGX | sulfonamides, urea derivatives | TCF7L2 | PGX |
| atenolol | DPYS | PGX | fluticasone propionate;montelukast | CA10 | PGX | sulindac | FMO3 | PGX |
| atenolol | GNB3 | PGX | fluvastatin | ACE | PGX | sumatriptan | GNB3 | PGX |
| atenolol;bisoprolol | PGX | fluvastatin | CETP | PGX | sunitinib | ABCB1 | PGX | |
| atenolol;hydrochlorothiazide | ALDH1A2 | PGX | fluvastatin | SLCO1B1 | PGX | sunitinib | FLT3 | PGX |
| atenolol;hydrochlorothiazide;metoprolol | SLC25A31 | PGX | fluvastatin;simvastatin | LIPC | PGX | tacrolimus | CYP3A5 | CPIC |
| atenolol;irbesartan | EDN1 | PGX | fluvoxamine | CYP2B6 | CPIC | tacrolimus | CYP3A5 | FDA |
| atenolol;metoprolol | ADRB2 | PGX | fluvoxamine | COMT | PGX | tacrolimus | CYP3A4 | PGX |
| atenolol;verapamil | CACNA1C | PGX | fluvoxamine | FGF2 | PGX | talinolol | ABCB1 | PGX |
| atomoxetine | CYP2D6 | CPIC | fluvoxamine | HTR2A | PGX | tamoxifen | CYP2D6 | CPIC |
| atomoxetine | CYP2D6 | DPWG | fluvoxamine | CYP2D6 | FDA | tamoxifen | CYP2D6 | DPWG |
| atomoxetine | CYP2D6 | FDA | fluvoxamine;milnacipran;paroxetine | HTR1A | PGX | tamoxifen | CYP2D6 | FDA |
| atomoxetine | SLC6A2 | PGX | fluvoxamine;paroxetine | HTR2A | PGX | tamoxifen | CYP19A1 | PGX |
| atorvastatin | APOE | PGX | folic acid | MTHFR | PGX | tamoxifen | UGT1A4 | PGX |
| atorvastatin | SLCO1B1 | PGX | folic acid;methotrexate | TLR4 | PGX | tamsulosin | CYP2D6 | FDA |
| atorvastatin;Bisphosphonates | FDPS | PGX | folic acid;vitamin b-complex, plain | MTHFR | PGX | taxanes | PGX | |
| atorvastatin;fluvastatin;hmg coa reductase inhibitors;lovastatin;pravastatin;simvastatin | HMGCR | PGX | folic acid;vitamin b-complex, plain | MTRR | PGX | tegafur | DPYD | DPWG |
| atorvastatin;hmg coa reductase inhibitors;pravastatin;simvastatin | HTR7 | PGX | follitropin beta;thyrotropin alfa;urofollitropin | FSHR | PGX | tegafur | DPYD | PGX |
| atorvastatin;hmg coa reductase inhibitors;rosuvastatin | COQ2 | PGX | forskolin | ABCB1 | PGX | telaprevir | CYP24A1 | PGX |
| atorvastatin;lovastatin;simvastatin | APOA5 | PGX | furosemide | ABCC4 | PGX | telmisartan | GNB3 | PGX |
| atorvastatin;pravastatin;simvastatin | ABCA1 | PGX | furosemide;spironolactone | ADD1 | PGX | telmisartan | SLCO1B3 | PGX |
| atorvastatin;quinapril | ACE | PGX | gabapentin | SLC7A5 | PGX | temozolomide | ABCB1 | PGX |
| atorvastatin;rifampin | SLCO1B1 | PGX | galantamine | CYP2D6 | FDA | tenofovir | ABCC4 | PGX |
| atorvastatin;simvastatin | PON1 | PGX | galantamine | CHAT | PGX | tenofovir disoproxil fumarate | CYP27B1 | PGX |
| atorvastatin;simvastatin | RYR2 | PGX | gefitinib | CYP2D6 | FDA | tenoxicam | CYP2C9 | CPIC |
| avatrombopag | CYP2C9 | FDA | gefitinib | ABCB1 | PGX | terbutaline | ADRB2 | PGX |
| axitinib;sorafenib | HIF1A | PGX | gefitinib | EGFR | PGX | tetrabenazine | CYP2D6 | FDA |
| azathioprine | NUDT15 | CPIC | geldanamycin | EGFR | PGX | thalidomide | ERCC1 | PGX |
| azathioprine | NUDT15 | DPWG | gemcitabine | NT5C2 | PGX | Thiazides, plain | GNB3 | PGX |
| azathioprine | TPMT | CPIC | gemcitabine | RRM1 | PGX | thiazolidinediones | AQP2 | PGX |
| azathioprine | TPMT | DPWG | gemcitabine;paclitaxel | CDA | PGX | thioguanine | NUDT15 | CPIC |
| azathioprine | TPMT | FDA | gemcitabine;paclitaxel | SLC29A1 | PGX | thioguanine | TPMT | CPIC |
| azathioprine | FTO | PGX | gemcitabine;Platinum compounds | CASP7 | PGX | thioguanine | TPMT | DPWG |
| azathioprine | NUDT15 | PGX | gemcitabine;Platinum compounds | RRM1 | PGX | thioguanine | TPMT | FDA |
| azathioprine;mercaptopurine | PGX | gemtuzumab ozogamicin | CD33 | PGX | thioridazine | CYP2D6 | FDA | |
| azathioprine;mercaptopurine;thioguanine | NUDT15 | PGX | glatiramer acetate | PGX | thioridazine | CYP2D6 | PGX | |
| azathioprine;purine analogues | ITPA | PGX | glibenclamide | NOS1AP | PGX | thiotepa | GSTP1 | PGX |
| belinostat | UGT1A1 | FDA | gliclazide | KCNJ11 | PGX | tianeptine | OPRM1 | PGX |
| benazepril | MTHFR | PGX | glimepiride | NOS1AP | PGX | ticagrelor | PEAR1 | PGX |
| benazepril;imidapril | AGT | PGX | glipizide | NOS1AP | PGX | timolol | PGX | |
| benazepril;perindopril | ACE | PGX | glucocorticoids | HSD3B1 | PGX | tiotropium | ADRB2 | PGX |
| Beta Blocking Agents | ADRB1 | PGX | granisetron | ABCB1 | PGX | tipifarnib | ABCB1 | PGX |
| Beta Blocking Agents | GRK5 | PGX | granisetron;palonosetron | ERCC1 | PGX | tipiracil hydrochloride;trifluridine | SLC29A1 | PGX |
| Beta blocking agents, selective | BST1 | PGX | haloperidol | CYP2D6 | DPWG | tocilizumab | IL6R | PGX |
| bevacizumab | ARMS2 | PGX | haloperidol | MC4R | PGX | tolbutamide | CYP2C9 | PGX |
| bevacizumab | CFH | PGX | haloperidol | SLC6A5 | PGX | tolterodine | CYP2D6 | FDA |
| bevacizumab | CXCR4 | PGX | halothane | CACNA1S | PGX | topiramate | GRIK1 | PGX |
| bevacizumab | EDN1 | PGX | hdl cholesterol;letrozole;triglycerides | CYP19A1 | PGX | topotecan | ABCG2 | PGX |
| bevacizumab;capecitabine;cetuximab;oxaliplatin | MGAT4A | PGX | Hepatitis vaccines | IL4R | PGX | tramadol | CYP2D6 | DPWG |
| bevacizumab;capecitabine;fluorouracil;irinotecan;leucovorin;oxaliplatin | VEGFA | PGX | heroin | OPRM1 | PGX | tramadol | CYP2D6 | FDA |
| bevacizumab;capecitabine;oxaliplatin | MGAT4A | PGX | heroin | SOD2 | PGX | tramadol | OPRM1 | PGX |
| bevacizumab;carboplatin;cisplatin;cyanocobalamin;folic acid;pemetrexed | MTHFR | PGX | heroin;methamphetamine | BDNF | PGX | trandolapril;verapamil | PTPRD | PGX |
| bevacizumab;fluorouracil;irinotecan;leucovorin | SHMT1 | PGX | heroin;opioids | OPRM1 | PGX | trastuzumab | ERBB2 | PGX |
| bevacizumab;pegaptanib;ranibizumab | VEGFA | PGX | highly active antiretroviral therapy (haart) | OPRM1 | PGX | trastuzumab | FCGR2A | PGX |
| bevacizumab;pemetrexed | GGH | PGX | hmg coa reductase inhibitors | LPA | PGX | triamcinolone | HCG22 | PGX |
| bevacizumab;ranibizumab | VEGFA | PGX | hmg coa reductase inhibitors | SLCO1B1 | PGX | trimipramine | CYP2C19 | CPIC |
| bilirubin | UGT1A | PGX | hmg coa reductase inhibitors;letrozole | CYP19A1 | PGX | trimipramine | CYP2D6 | CPIC |
| bisantrene | ABCB1 | PGX | hmg coa reductase inhibitors;simvastatin | GATM | PGX | trimipramine | CYP2D6 | FDA |
| bisoprolol | ACY3 | PGX | hormonal contraceptives for systemic use | F5 | PGX | troglitazone | PGX | |
| Bisphosphonates | ABCC4 | PGX | Hydralazine-isosorbide dinitrate | GNB3 | PGX | tropisetron | CYP2D6 | CPIC |
| Bisphosphonates | FDPS | PGX | hydrochlorothiazide | ADD1 | PGX | Tumor necrosis factor alpha (TNF-alpha) inhibitors | IL23R | PGX |
| Bisphosphonates;clodronate;etidronic acid;risedronate;tiludronate | IL1B | PGX | hydrochlorothiazide | KCNJ1 | PGX | Tumor necrosis factor alpha (TNF-alpha) inhibitors | TNF | PGX |
| Bisphosphonates;pamidronate;zoledronate | CYP2C8 | PGX | hydrocodone | OPRM1 | PGX | Tumor necrosis factor alpha (TNF-alpha) inhibitors;ustekinumab | IL1B | PGX |
| bleomycin;cisplatin;etoposide | BLMH | PGX | hydroxyurea | MAP3K5 | PGX | ustekinumab | TIRAP | PGX |
| bleomycin;cisplatin;etoposide | GSTP1 | PGX | ibuprofen | PTGS2 | PGX | valbenazine | CYP2D6 | FDA |
| bleomycin;cyclophosphamide;doxorubicin;prednisone;rituximab;vincristine;vindesine | ABCB1 | PGX | idarubicin | NCF4 | PGX | valganciclovir | ABCB1 | PGX |
| boceprevir;peginterferon alfa-2a;peginterferon alfa-2b;ribavirin | IFNL3;IFNL4 | PGX | idarubicin | RAC2 | PGX | valproic acid | POLG | PGX |
| bortezomib;dexamethasone;melphalan;prednisone | CTLA4 | PGX | iloperidone | CYP2D6 | FDA | valproic acid | UGT1A6 | PGX |
| botulinum toxin type a | TRPV1 | PGX | iloperidone | CYP2D6 | PGX | vancomycin | PGX | |
| brexpiprazole | CYP2D6 | DPWG | imatinib | CYP2B6 | PGX | vardenafil | CYP3A5 | PGX |
| brexpiprazole | CYP2D6 | FDA | imatinib | EGFR | PGX | varenicline | CHRNA4 | PGX |
| brivaracetam | CYP2C19 | FDA | imatinib | SLC22A1 | PGX | venlafaxine | CYP2D6 | DPWG |
| bucindolol | ADRB1 | PGX | imatinib | SLC22A4 | PGX | venlafaxine | CYP2D6 | FDA |
| budesonide | PGX | imipramine | CYP2C19 | CPIC | venlafaxine | HTR2A | PGX | |
| budesonide;flunisolide;fluticasone propionate;glucocorticoids;nedocromil;triamcinolone | GLCCI1 | PGX | imipramine | CYP2C19 | DPWG | verapamil | KCNIP1;KCNMB1 | PGX |
| bufuralol;dextromethorphan | CYP2D6 | PGX | imipramine | CYP2D6 | CPIC | vincristine | ABCB1 | PGX |
| bumetanide;furosemide;torasemide | SLC12A3 | PGX | imipramine | CYP2D6 | DPWG | vincristine | CEP72 | PGX |
| buprenorphine | COMT | PGX | imipramine | CYP2D6 | FDA | vitamin e | CYP4F2 | PGX |
| buprenorphine | OPRD1 | PGX | imiquimod | TLR7 | PGX | Vitamin K | CYP4F2 | PGX |
| buprenorphine;fentanyl;meperidine;morphine;opioids;pentazocine | PGX | indinavir | CYP3A4 | PGX | voriconazole | CYP2C19 | CPIC | |
| buprenorphine;fentanyl;tramadol | COMT | PGX | indinavir | UGT1A1 | PGX | voriconazole | CYP2C19 | DPWG |
| bupropion | GALR1 | PGX | infliximab | TNF | PGX | voriconazole | CYP2C19 | FDA |
| bupropion;Drugs used in nicotine dependence;nicotine;varenicline | CHRNA5 | PGX | infliximab;Tumor necrosis factor alpha (TNF-alpha) inhibitors | TNFRSF1B | PGX | voriconazole | CYP3A4 | PGX |
| bupropion;nicotine | DRD1 | PGX | insulin recombinant;zinc acetate | SLC30A8 | PGX | vortioxetine | CYP2D6 | FDA |
| bupropion;nicotine;varenicline | CHRNA5 | PGX | interferon alfa-2a, recombinant;interferon alfa-2b, recombinant;ribavirin | FTO | PGX | warfarin | CYP2C9 | DPWG |
| busulfan | CYP2C9 | PGX | interferon alfa-2a, recombinant;peginterferon alfa-2b | IDO1 | PGX | warfarin | CYP2C9 | FDA |
| busulfan;cyclophosphamide | CTH | PGX | interferon alfa-2b, recombinant;ribavirin | ITPA | PGX | warfarin | CYP4F2 | FDA |
| butorphanol | COMT | PGX | interferon beta-1a | RORA | PGX | warfarin | VKORC1 | FDA |
| caffeine | ADORA2A | PGX | interferon beta-1a;interferon beta-1b | HLA-DQA1 | PGX | warfarin | PGX | |
| caffeine | CYP1A2 | PGX | interferon beta-1a;interferon beta-1b | IRF6 | PGX | warfarin | CYP4F2 | PGX |
| calcein | ABCB1 | PGX | interferons | IFNL3;IFNL4 | PGX | warfarin | VKORC1 | PGX |
| calcein;lopinavir | ABCC2 | PGX | interferons | STAT3 | PGX | XK469 | AOX1 | PGX |
| calcium | VDR | PGX | interferons;peginterferon alfa-2a;peginterferon alfa-2b;ribavirin | IFNL3 | PGX | zileuton | PRORP | PGX |
| calcium channel blockers | CACNA1C | PGX | interferons;ribavirin | IL18 | PGX | zoledronate | FDPS | PGX |
| calcium channel blockers;nitrendipine | KCNH2 | PGX | irbesartan | CYP2C9 | PGX | zuclopenthixol | CYP2D6 | DPWG |
| candesartan | AGTR1 | PGX | irinotecan | UGT1A1 | DPWG | |||
| cangrelor | P2RY12 | PGX | irinotecan | UGT1A1 | FDA | |||
| cannabinoids | FAAH | PGX | irinotecan | UGT1A1 | PGX | |||
| cannabinoids;cocaine;ethanol;nicotine;opioids | OPRM1 | PGX | irinotecan;raltitrexed | TYMS | PGX | |||
| capecitabine | DPYD | CPIC | isoflurane | CACNA1S | PGX | |||
| capecitabine | DPYD | FDA | isoniazid | BACH1 | PGX | |||
| capecitabine | DPYD | PGX | isoniazid;phenytoin | NAT2 | PGX | |||
| capecitabine | TYMS | PGX | isoniazid;pyrazinamide;rifampin | NAT2 | PGX | |||
| capecitabine;docetaxel | CYP1A1 | PGX | isoniazid;rifampin | GSTP1 | PGX | |||
| capecitabine;epirubicin;platinum | GSTP1 | PGX | isoproterenol | ADRB2 | PGX | |||
| capecitabine;fluorouracil | HLA-G | PGX | ivacaftor | CFTR | PGX | |||
| capecitabine;fluorouracil;leucovorin;oxaliplatin | MTHFR | PGX | ivacaftor-lumacaftor | CFTR | PGX | |||
| capecitabine;fluorouracil;tegafur | TYMS | PGX | ivacaftor-tezacaftor | CFTR | PGX | |||
| capecitabine;oxaliplatin | PTGS2 | PGX | ketorolac | PGX | ||||
| capecitabine;radiotherapy | AREG | PGX | l-methylfolate;Vitamin B-complex, Incl. Combinations | MTHFR | PGX | |||
| captopril | ACE | PGX | l-phenylalanine | PAH | PGX | |||
| carbamazepine | HLA-A | CPIC | l-tryptophan | SLC22A2 | PGX | |||
| carbamazepine | HLA-A | FDA | lamivudine;lopinavir;ritonavir;zidovudine | ABCB1 | PGX | |||
| carbamazepine | HLA-B | CPIC | lamotrigine | UGT1A4 | PGX | |||
| carbamazepine | HLA-B | FDA | lansoprazole | CYP2C19 | CPIC | |||
| carbamazepine | CYP3A5 | PGX | lansoprazole | CYP2C19 | DPWG | |||
| carbamazepine | SCN1A | PGX | lansoprazole;omeprazole;rabeprazole | IL1B | PGX | |||
| carbamazepine;clobazam;ethosuximide;lamotrigine;levetiracetam;oxcarbazepine;valproic acid | SCN1A | PGX | lansoprazole;tacrolimus | ABCB1 | PGX | |||
| carbamazepine;oxcarbazepine | SCN1A | PGX | lapatinib | HLA-DQA1 | FDA | |||
| carbamazepine;phenobarbital;phenytoin;valproic acid | CYP1A1 | PGX | lapatinib | HLA-DRB1 | FDA | |||
| carbamazepine;phenytoin;valproic acid | GABRA1 | PGX | latanoprost | ABCC4 | PGX | |||
| carbocisteine | PAH | PGX | ledipasvir;sofosbuvir | IFNL3;IFNL4 | PGX | |||
| carboplatin;cisplatin | EIF3A | PGX | leflunomide | ESR1 | PGX | |||
| carboplatin;cisplatin;docetaxel;erlotinib;gefitinib;paclitaxel | MAP3K1 | PGX | lenalidomide;thalidomide | CTNNB1 | PGX | |||
| carboplatin;cisplatin;docetaxel;gemcitabine;paclitaxel | RAF1 | PGX | letermovir | SLCO1B1 | PGX | |||
| carboplatin;cisplatin;docetaxel;paclitaxel | PGX | letrozole | ESR1 | PGX | ||||
| carboplatin;cisplatin;fluorouracil | SLC19A1 | PGX | levodopa | HOMER1 | PGX | |||
| carboplatin;cisplatin;gemcitabine | GPX5 | PGX | levodopa | SLC6A3 | PGX | |||
| carboplatin;cisplatin;oxaliplatin;platinum;Platinum compounds | ERCC1 | PGX | liraglutide | GLP1R | PGX | |||
| carboplatin;cyclophosphamide;thiotepa | ALDH1A1 | PGX | lisinopril | ACE | PGX | |||
| carboplatin;docetaxel;paclitaxel | BCL2 | PGX | lithium | ASIC2 | PGX | |||
| carboplatin;docetaxel;trastuzumab | ERBB3 | PGX | lithium;valproic acid | PGX | ||||
| carboplatin;gemcitabine | CYP2C8 | PGX | lofexidine | CYP2D6 | FDA | |||
| carboplatin;lonafarnib;paclitaxel | FNTB | PGX | lopinavir | SLCO1B1 | PGX | |||
| carboplatin;paclitaxel | MAD1L1 | PGX | lorazepam | UGT2B15 | PGX | |||
| carboplatin;paclitaxel | SLCO1B3 | PGX | losartan | STK39 | PGX | |||
| carboplatin;taxanes | GSR | PGX | lovastatin | LDLR | PGX | |||
| carisoprodol | CYP2C19 | FDA | lovastatin acid | SLCO1B1 | PGX | |||
| carvedilol | CYP2D6 | FDA | lumacaftor | CFTR | PGX | |||
| carvedilol | ADRB2 | PGX | lumefantrine | CYP3A4 | PGX | |||
| catecholamines | ADRB1 | PGX | lurasidone | HTR1A | PGX | |||
| catecholamines;metformin | SLC22A3 | PGX | Measles vaccines | TLR3 | PGX | |||
| cavosonstat | CFTR | PGX | meclizine | CYP2D6 | FDA | |||
| cefotaxime | SLC22A8 | PGX | meloxicam | CYP2C9 | CPIC | |||
| ceftriaxone | ABCG2 | PGX | melphalan | SLC7A5 | PGX | |||
| celecoxib | CYP2C9 | FDA | memantine | NR1I2 | PGX | |||
| celecoxib | ALOX12 | PGX | mephenytoin | CYP2C19 | PGX | |||
| cerivastatin | SLCO1B1 | PGX | mercaptopurine | NUDT15 | CPIC | |||
| cetuximab | EGF | PGX | mercaptopurine | TPMT | CPIC | |||
| cetuximab;irinotecan;leucovorin;tegafur | EGFR | PGX | mercaptopurine | TPMT | FDA | |||
| cetuximab;irinotecan;panitumumab | AREG | PGX | mercaptopurine | FTO | PGX | |||
| cetuximab;panitumumab | KRAS | PGX | mercaptopurine | NUDT15 | PGX | |||
| cevimeline | CYP2D6 | FDA | mercaptopurine;methotrexate | ITPA | PGX | |||
| chlorothiazide;hydrochlorothiazide | PGX | metformin | SLC22A1 | PGX | ||||
| chlorpromazine;clozapine;haloperidol;olanzapine;quetiapine;risperidone;trifluoperazine | EPM2A | PGX | metformin | SLC47A1 | PGX | |||
| chlorthalidone;lisinopril | MMP3 | PGX | metformin;sulfonamides, urea derivatives | KCNJ11 | PGX | |||
| cisplatin | ACYP2 | PGX | methacholine | ADRB2 | PGX | |||
| cisplatin | ERCC1 | PGX | methadone | ABCB1 | PGX | |||
| cisplatin;cyclophosphamide | CYP2E1 | PGX | methadone | CYP3A4 | PGX | |||
| cisplatin;cyclophosphamide | GSTM3 | PGX | methamphetamine | FAAH | PGX | |||
| cisplatin;cyclophosphamide;doxorubicin;methotrexate;vincristine | ABCC3 | PGX | methotrexate | ATIC | PGX | |||
| cisplatin;fluorouracil | GALNT14 | PGX | methotrexate | MTHFR | PGX | |||
| cisplatin;fluorouracil;mitoxantrone | GALNT14 | PGX | methotrexate | SLC19A1 | PGX | |||
| cisplatin;fluorouracil;oxaliplatin | VEGFA | PGX | methoxyflurane | CACNA1S | PGX | |||
| cisplatin;fluorouracil;radiotherapy | MUTYH | PGX | methylphenidate | CES1 | PGX | |||
| cisplatin;gemcitabine | RRM1 | PGX | methylphenidate | DRD2 | PGX | |||
| cisplatin;irinotecan | UBE2I | PGX | methylphenidate | SLC6A2 | PGX | |||
| cisplatin;oxaliplatin;Platinum compounds | GSTP1 | PGX | methylprednisolone;prednisolone | ABCB1 | PGX | |||
| cisplatin;oxaliplatin;platinum;Platinum compounds | ERCC2;KLC3 | PGX | metoclopramide | CYP2D6 | FDA | |||
| cisplatin;pemetrexed | ABCC2 | PGX | metoprolol | CYP2D6 | DPWG | |||
| citalopram | CYP2C19 | CPIC | metoprolol | CYP2D6 | FDA | |||
| citalopram | CYP2C19 | DPWG | metoprolol | ADRB1 | PGX | |||
| citalopram | CYP2C19 | FDA | midazolam | POR | PGX | |||
| citalopram | HTR2A | PGX | migalastat | GLA | PGX | |||
| citalopram;escitalopram | PGX | milnacipran | HTR1A | PGX | ||||
| citalopram;fluoxetine | GSK3B | PGX | mirabegron | CYP2D6 | FDA | |||
| citalopram;fluoxetine;paroxetine;sertraline | REEP5 | PGX | mirtazapine | FKBP5 | PGX | |||
| cladribine;cytarabine | RRM1 | PGX | mirtazapine;venlafaxine | TPH2 | PGX | |||
| cladribine;fluorouracil;gemcitabine | NT5C1A | PGX | mitotane | CYP2B6 | PGX | |||
| clobazam | CYP2C19 | FDA | mivacurium | BCHE | FDA | |||
| clodronate | VDR | PGX | modafinil | ABCB1 | PGX | |||
| clomipramine | CYP2C19 | CPIC | montelukast | LTA4H | PGX | |||
| clomipramine | CYP2C19 | DPWG | morphine | KCNJ6 | PGX | |||
| clomipramine | CYP2D6 | CPIC | morphine | OPRM1 | PGX | |||
| clomipramine | CYP2D6 | DPWG | morphine;nortriptyline | ABCB1 | PGX | |||
| clomipramine | CYP2D6 | FDA | muraglitazar | EDN1 | PGX | |||
| clomipramine;liothyronine;lithium;nefazodone;venlafaxine | HTR1B | PGX | mycophenolate mofetil | SLCO1B3 | PGX | |||
| clomipramine;lithium;nefazodone;paroxetine;venlafaxine | ABCB1 | PGX | mycophenolic acid | SLCO1B3 | PGX | |||
| clomipramine;nefazodone;paroxetine;venlafaxine | FKBP5 | PGX | naloxone | OPRM1 | PGX | |||
| clonidine | GNB3 | PGX | naltrexone | DBH | PGX | |||
| clopidogrel | CYP2C19 | CPIC | naproxen | CYP2C9 | PGX | |||
| clopidogrel | CYP2C19 | DPWG | nateglinide | SLCO1B1 | PGX | |||
| clopidogrel | CYP2C19 | FDA | nebivolol | CYP2D6 | FDA | |||
| clopidogrel | CES1 | PGX | nelfinavir | CYP2C19 | PGX | |||
| clozapine | CYP2D6 | FDA | nemonapride | ANKK1 | PGX | |||
| clozapine | ABCB1 | PGX | nevirapine | CYP2B6 | PGX | |||
| clozapine | HTR2C | PGX | nicotine | CHRNA3 | PGX | |||
| clozapine | NTRK2 | PGX | nicotine | CHRNA5 | PGX | |||
| clozapine;olanzapine | MC4R | PGX | nicotine;Opium alkaloids and derivatives | CHRNA5 | PGX | |||
| clozapine;olanzapine;risperidone | DRD2 | PGX | nifedipine | SLC14A2 | PGX | |||
| cocaine | DRD2 | PGX | nilotinib | UGT1A1 | FDA | |||
| cocaine | OPRM1 | PGX | nimodipine | CACNA1C | PGX | |||
| cocaine;ethanol;nicotine | OPRM1 | PGX | nitrendipine | AGTR1 | PGX | |||
| cocaine;heroin | OPRM1 | PGX | nitroprusside | ACE | PGX | |||
| codeine | CYP2D6 | CPIC | nitrous oxide | MTHFR | PGX | |||
| codeine | CYP2D6 | DPWG | nortriptyline | CYP2D6 | CPIC | |||
| codeine | CYP2D6 | FDA | nortriptyline | CYP2D6 | DPWG | |||
| codeine | OPRM1 | PGX | nortriptyline | CYP2D6 | FDA | |||
| conjugated estrogens | SULT1A1 | PGX | nortriptyline | ABCB1 | PGX | |||
| conjugated estrogens;medroxyprogesterone | VDR | PGX | nortriptyline | GNB3 | PGX | |||
| corticosteroids | BCL2L11 | PGX | ||||||
| corticosteroids | CRHR1 | PGX | ||||||
| corticosteroids;selective beta-2-adrenoreceptor agonists | ADRB2 | PGX | ||||||
| cotinine | CHRNA5 | PGX | ||||||
| cotinine glucuronide | UGT1A9 | PGX | ||||||
| cotinine;nicotine | OPRM1 | PGX | ||||||
| coumarin | CYP2A6 | PGX | ||||||
| creatine | MOBP | PGX | ||||||
| curcumin;ivacaftor | CFTR | PGX | ||||||
| cyclophosphamide | ABCC4 | PGX | ||||||
| cyclophosphamide | CYP2C19 | PGX | ||||||
| cyclophosphamide | SOD2 | PGX | ||||||
| cyclophosphamide;cytarabine;daunorubicin;mercaptopurine;methotrexate;prednisone;vincristine | DROSHA | PGX | ||||||
| cyclophosphamide;dexamethasone;lenalidomide;thalidomide | CTNNB1 | PGX | ||||||
| cyclophosphamide;docetaxel | VEGFA | PGX | ||||||
| cyclophosphamide;docetaxel;doxorubicin;epirubicin;fluorouracil | SLCO1B1 | PGX | ||||||
| cyclophosphamide;doxorubicin | CYP2C19 | PGX | ||||||
| cyclophosphamide;doxorubicin;fluorouracil | ATM | PGX | ||||||
| cyclophosphamide;doxorubicin;fluorouracil | SLC22A16 | PGX | ||||||
| cyclophosphamide;doxorubicin;fluorouracil;methotrexate | NOS3 | PGX | ||||||
| cyclophosphamide;doxorubicin;prednisone;rituximab;vincristine | GSTA1 | PGX | ||||||
| cyclophosphamide;epirubicin | GSTP1 | PGX | ||||||
| cyclophosphamide;epirubicin;fluorouracil | NQO1 | PGX | ||||||
| cyclophosphamide;epirubicin;fluorouracil | PIGB | PGX | ||||||
| cyclophosphamide;epirubicin;paclitaxel | CYP1B1 | PGX | ||||||
| cyclophosphamide;fluorouracil;methotrexate | FGFR4 | PGX | ||||||
| cyclosporine | CYP3A4 | PGX | ||||||
| cyclosporine | CYP3A5 | PGX | ||||||
| cyclosporine;dicloxacillin | ABCB1 | PGX | ||||||
| cyclosporine;mycophenolate mofetil | IL10 | PGX | ||||||
| cyclosporine;mycophenolate mofetil;sirolimus;tacrolimus | UGT1A8 | PGX | ||||||
| cyclosporine;mycophenolic acid | ABCC2 | PGX | ||||||
| cyclosporine;sirolimus;tacrolimus | TCF7L2 | PGX | ||||||
| cyclosporine;tacrolimus | CRTC2 | PGX | ||||||
| cysteamine | CFTR | PGX | ||||||
| cytarabine | CDA | PGX | ||||||
| cytarabine;daunorubicin;dexrazoxane;idarubicin | ABCB1 | PGX | ||||||
| cytarabine;daunorubicin;etoposide;mitoxantrone | SLCO1B1 | PGX | ||||||
| cytarabine;fludarabine;gemtuzumab ozogamicin;idarubicin | CYP2E1 | PGX | ||||||
| cytarabine;idarubicin | NT5C3A | PGX |
There you have it! With information, free is not always good and useful. A lot of users come to Xcode after having tried Promethease for $5. If you just want to satisfy your curiosity and not really looking for anything specific, then Promethease may be the way to go. But, if you want specific, organized, and actionable insights from your genetic data about your health and wellbeing, then you certainly must try the Xcode Health reports!
Updated on January 15th, 2022
GEDmatch is primarily a free website and a powerful genealogy tool that lets users upload their DNA raw data files to find related individuals. It offers a bunch of free tools as well as some paid tools. Read on to know how to get started with GEDmatch for in-depth research about your family history!
GEDmatch is a free genealogy website where users can upload their DNA raw data files to track down family members and learn more about their family history.
More reading: What is a DNA raw data file?
Cofounded by Curtis Rogers and John Olson in 2010, it is now owned by Verogen and is based in San Diego, CA. With a global genealogical database and 45+ genetic tools, GEDmatch focuses on genealogy research to provide accessible and effective data to its consumers.
GEDmatch allows you to compare your DNA raw data using a variety of easy-to-use applications, most of which are free. The services include 45+ genetic tools which help understand and explore your DNA and ancestry.
The following are the free tools offered by GEDMatch:
The One-to-Many DNA Comparison tool can be a great place to start. This tool provides a list of people that you share chromosome segments with, along with their email IDs for quick contact. You can also sort and filter your matches.
"Total cM" (centimorgans) refers to the total length of all substantial DNA segments you share with a DNA match. The larger the number of centimorgans you share with another user, the more closely you are related.
"Overlap" is the total number of SNPs or single nucleotide polymorphisms that you share with your match.
A one-to-one autosomal DNA comparison enables you to make detailed comparisons of two DNA profiles. The results include a list of shared chromosomes and their locations on the genome. You can view your results in tabular or graphical format, or both.
This tool allows you to see the percentage of each ethnicity in your DNA, displayed as a simple pie chart. There are also different calculators which provide specialized results for specific ethnicities.
There are 7 projects to choose from in the Admixture (Heritage) tool.
With this tool, you can examine segment matches between several individuals at the same time (up to 10). You can quickly identify shared common segments, which may indicate a single common ancestor for multiple people. The graphics used to display the results may not work in certain browsers.
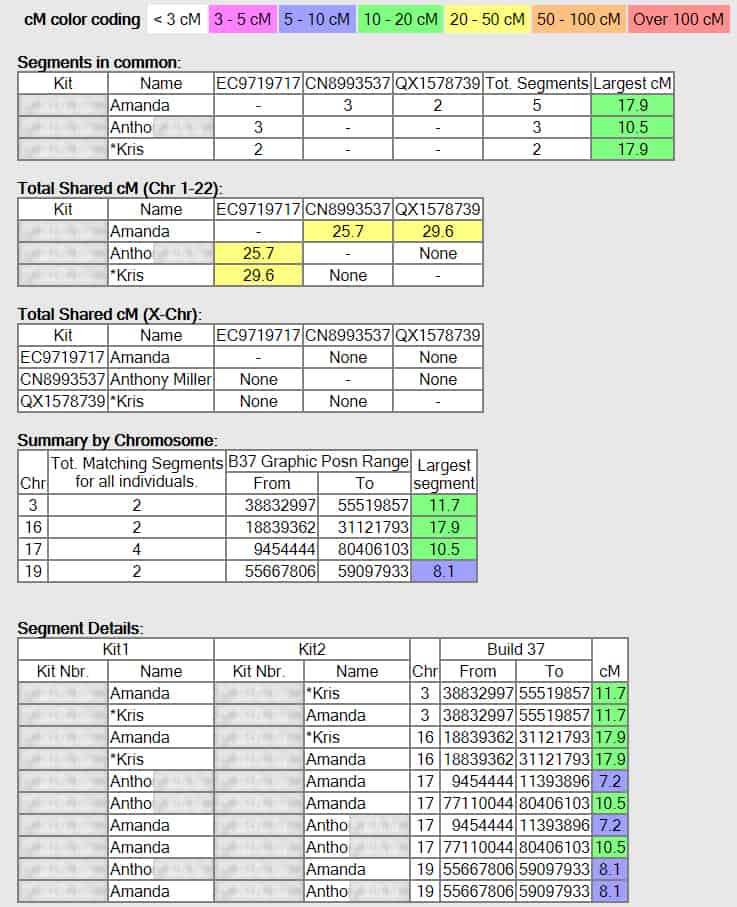
Segment Search: With this tool, you can search your top matches for overlapping segments. This can help you identify matches that share the most recent common ancestors.
Q-Matching Enhanced One-to-One: This tool is similar to the one-to-one comparison tool and compares your DNA with the DNA of a match. Additionally, it examines the quality of the shared segments.
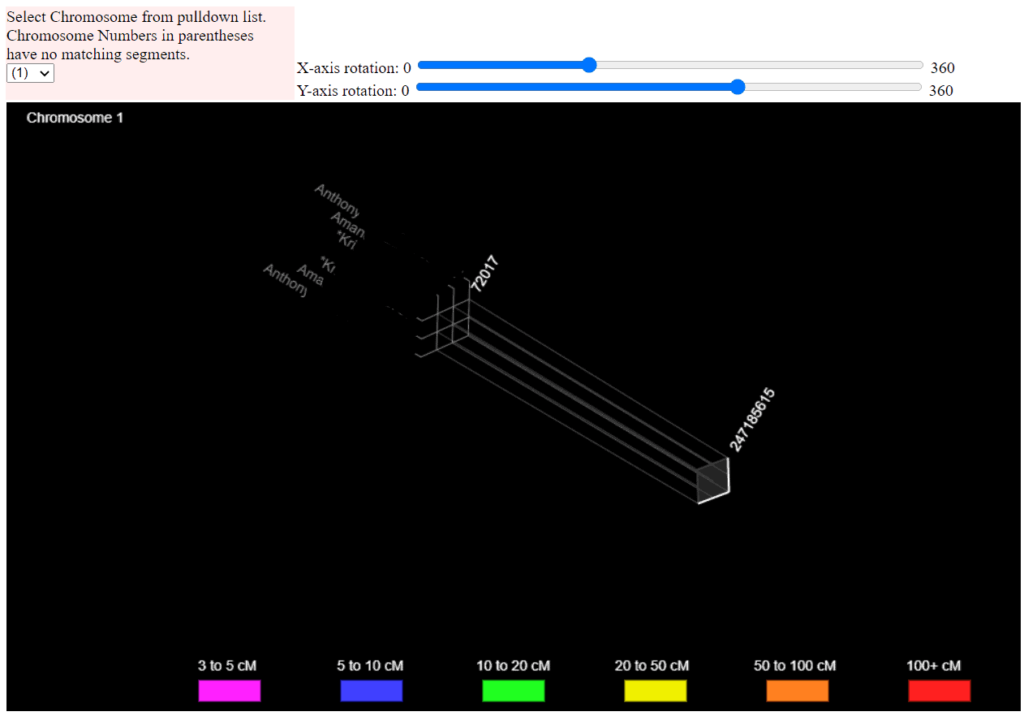
Phasing Tool: It attempts to separate your paternal and maternal autosomal DNA.
Cluster Tool: This is a sorting feature that groups your matches into clusters based on the degree of similarity, indicating descent from a recent common ancestor."
These Tier 1 tools enable complex genealogy research and help investigate your DNA at a much more advanced level.
The page also displays the following information for signed-in users:
GEDmatch is predominantly free, with an option to pay for access to more advanced tools.
Three basic genealogy analysis tools (one-to-one, one-to-many, and admixture), along with 45+ other genealogic analysis tools, are available for free on your account.
Apart from these, you can gain access to more complex tools from the Tier 1 subscription priced at $10/month. The Tier 1 plan offers a choice between auto-renewal and an on-demand payment option.
GEDmatch collects your name, an optional alias, and email address for your registration. Once registered, other personal information such as your sex, genetic information, genealogy data, and payment information can be provided with consent.
GEDmatch automatically collects certain information regarding visitors to their site, solely for internal purposes, such as site improvement. This includes your IP address, browsing actions, information about your equipment, and usage patterns.
The site may use third-party tools to help understand statistical data about its visitors. GEDmatch does not reveal any personal information to these tools.
When you upload your DNA to GEDmatch, you can opt for one of the four privacy options:
Private: DNA data is not available to other people.
Public + opt-in: DNA data is available for one-to-many searches, including law enforcement searches.
Public + opt-out: DNA data is available for comparison to any raw data in the GEDmatch database, except law enforcement searches.
Research: DNA data is available for one-to-one comparison with public or research DNA. It does not allow one-to-many searches.
GEDmatch confirmed a breach in data privacy in July 2020, after genetic information was made available to the police. For further information, you can read GEDmatch's Privacy Policy.
GEDmatch is a standard tool that is recommended among various genealogy groups on Facebook. It has also proved to be useful for law enforcement investigations to solve crimes. GEDmatch helped with the arrest of the Golden State Killer, responsible for more than 50 rapes and at least 13 murders in California in the 1970s and 1980s. Ever since this case was solved, GEDmatch has transformed into a popular crime-solving tool.
The FBI and the police in California worked with a professional genealogist, Barbara Rae, to match DNA samples from the crime scene to GEDmatch profiles of the killer's third and fourth cousins.
Since then, GEDmatch has helped solve around 70 violent crimes. However, the site's new prominence as a crime-solving tool has divided genealogists concerned about this practice invading user privacy and those keen to work with law enforcement.
Genetic Lifehacks is a website where users can learn about the genetic connection between their diet and health. Founded by Debbie Moon in 2015, the company aims to combine an engineering mindset with biological systems and apply genetics to dietary and lifestyle choices. Genetic Lifehacks claims to use people's DNA to optimize their diets and lifestyles, based on their exact needs.
Genetic Lifehacks offers the following products and services:
The website offers a "Top 5 Topics" report customized based on vital topics for each individual. It spans over 11-15 pages and includes the following:
-Explanation of the topic
-Discussion of the genetic variants
This report analyzes 20+ genes and explains how these genes influence sleep aspects like melatonin secretion, restless leg syndrome, insomnia, and caffeine response.
The report includes the following:
Additionally, it includes information on circadian rhythm and homeostatic sleep drive.
Xcode Life's Gene Sleep Report - 15 traits for $20
This cheat sheet quickly shows you which article on the website matches your gene results. It is organized into the following categories:
Each category has a short article description, the risk gene, and a color-coded section for the individual genome. It also explains the different interactions of gene variants from different article sections.
Source: Genetic Life Hacks
This free introductory course explains what genetic data means. It is a 4-day course, providing essential information on genetics.
However, please bear in mind that Debbie Moon is not a doctor.
She only provides information and opinions, curated using peer-reviewed research and statistics. Individuals must seek assistance from a doctor for any medical advice regarding the provided information.
Once you subscribe to a membership plan and purchase a product, you can upload your raw DNA data file from 23andMe or AncestryDNA on the checkout page. The upload process takes about a minute. Once that is done, you can complete the checkout.
After checkout, you will receive an email with your Top 5 Report within 1-3 business days.
Genetic Lifehacks offers a membership option to its users at the following rates:
Genetic Lifehacks offers a range of other services for its users.
According to their website, Genetic Lifehacks does not store personal information except when purchasing a product.
The website collects user activity statistics and cookies to improve user experience. No tracking software is used on the website, ensuring user privacy.
If one chooses to post their personal information like name and email in the comments section, it gets recorded on the site.
Genetic Lifehacks stores user genetic data offline on an encrypted drive. It is deleted after 30 days and not used for research. The information is used only for consultation and not shared with third parties.
For further information, read the Genetic Lifehacks Privacy Policy.
You can learn more in the News and Research section of Genetic Lifehacks.
Trendhunter reviewed Genetic Lifehacks and scored it a 4.9.
| Genetic Lifehacks | Xcode Life | |
| Type of genetic testing | DNA raw data analysis | DNA raw data analysis |
| Ancestry reports | Not offered | Not offered |
| Health reports | Not offered | Offered |
| Pharmacogenomic reports | Not offered | Offered |
| Raw data upload | Only from 23andMe and AncestryDNA | DNA raw data from all major providers accepted. Comprehensive list |
| Number of reports | 5 topics | 13 |
| Price | Annual membership- $39Other services - $29-$99 | Reports: $10-$40Bundles: $49-$99Price details |
| Free tools | Nil | 3 free tools (Gene Tool, DNA raw data merge, DNA raw data convert) available for all |
| Sample reports | Nil | Available as detailed report walkthrough videos |
Gene By Gene was co-founded in 2000 by Bennett Greenspan and Max Blankfeld, with its headquarters in Houston, Texas. Currently, the company is headed by Dr. Lior Rauchberger as part of a merger with a leading Australian genomics firm, myDNA. The company’s labs are CAP, CLIA, NYSDoH, CA, and AABB accredited.
Gene By Gene laid the foundations of DNA-based ancestry and genealogy as FamilyTreeDNA (FTDNA). The first company of its kind, FTDNA, collaborated with National Geographic’s Genographic Project as their testing provider.
The company was the first lab to offer DNA-based ancestry and genealogy services as its subsidiary, FamilyTreeDNA, since 2000. It has now expanded its services to clinical, forensic, and research genetics. Amid the COVID-19 pandemic, Gene By Gene has opened its door for COVID-19 testing services too. The company caters to a wide range of individuals - clinicians, consumers, and researchers - worldwide.
For starters, clinicians, consumers, and researchers alike can register with the company. Upon registration, they can browse from a list of tests to place an order. The account dashboard has five tabs for browsing.
Clinicians can add as many patient profiles as they want.
Clinicians can opt for a carrier screening test for other members of the patients’ families, too, if needed.
Gene By Gene’s clinical testing services includes the following:
The clinical testing services of Gene By Gene can be availed either as a healthcare provider (for your patient) or a consumer.
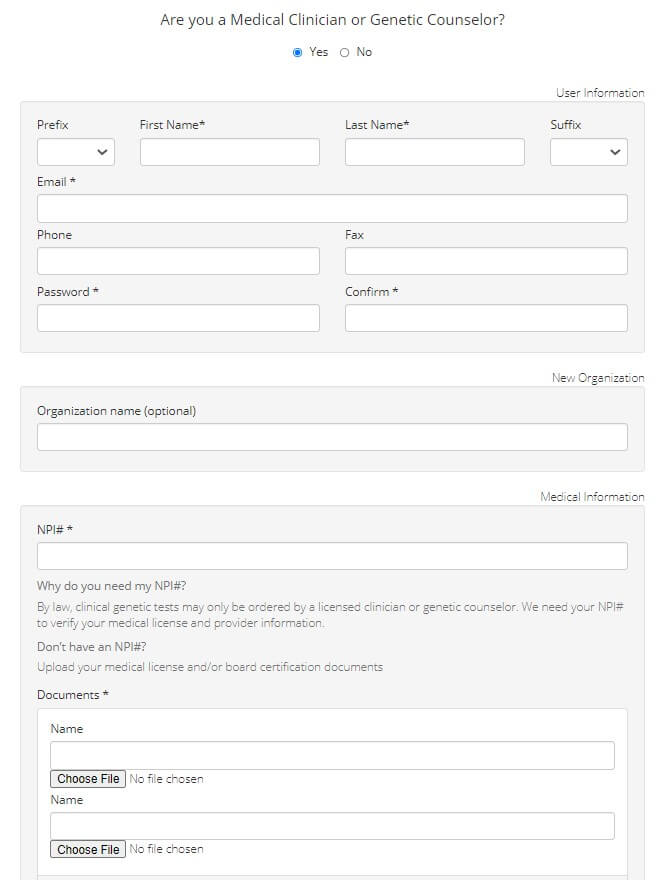
Image: Registration for genetic counselors/medical clinicians
The company provides Investigative Genetic Genealogy (IGG) services to law enforcement organizations through its subsidiary, FamilyTreeDNA. The company has its in-house lab and many genetic genealogists who discreetly identify individuals. They do so by matching them to unknown samples of DNA from cases involving homicides or sexual assault. This is brought about by familial matching within the FamilyTreeDNA database.
Law enforcement officials can submit their case along with evidence for review to the company. The staff reviews the case and checks for sample suitability before proceeding with testing.
Gene By Gene offers a wide range of DNA testing services, including paternity, maternity, immigration, and legal testing.
Gene By Gene offers a range of sequencing and microarray services for researchers from academic institutions, pharma companies, and hospital and medical research centers. The labs are CAP/CLIA accredited, and services are HIPAA compliant. Services provided are:
The lab uses Illumina NovaSeq 6000 for WGS and WES services. The data is provided as FASTQ. For Fragile X service, carrier status is checked using AmplideX, FMR1.
Gene By Gene offers its ancestry testing service through its DTC (direct-to-consumer) genetic testing subsidiary, FamilyTreeDNA. This service has an entirely separate website from where clients can access Ancestry Testing's services. The consumer can sign up and start ordering kits with the specific service.
The different DNA tests offered are:
A part of the Family Finder test, this feature gives a percentile breakdown of the populations to which your autosomal DNA is connected. It can be accessed either from the navigation bar or from the consumer login dashboard.
This feature has two tabs:
The three tests (Family Finder, Y-DNA, and mtDNA) have separate databases of people who undertake each test. Therefore, upon a consumer doing either of the three tests, his data are matched with people who had undertaken the test before and checked for common ancestral descent. This information builds the base for the share origins tab of the myOrigins feature.
Gene By Gene provides a comprehensive carrier screening service that includes panel tests with 253 autosomal and X-linked recessive conditions (all conditions included by the American College of Medical Genetics and American Congress of Obstetricians and Gynecologists). A simple cheek swab collects the sample for the screening test. The test results are ready in 2 weeks.
The company also provides complimentary genetic counseling services upon reception of carrier screening results. The service can be directly accessed by contacting Gene By Gene and scheduling an appointment.
Image: Snippet of Carrier Status Report
Xcode Life’s Carrier Status Report - 360+ Traits, $40 - No DNA test required
This test is used to determine the alpha tryptase copy number. It can be requested by healthcare providers, researchers, and consumers to check for serum tryptase levels. Suppose consumers order a test themselves; in that case, the test request form must be approved and signed by the consumer’s healthcare provider.
* Note: Alpha tryptase is produced by the gene TPSAB1 and beta tryptase by genes TPSAB1 and TPSAB2. Individuals with one copy of inherited α-tryptase on the gene sequence have normal serum tryptase levels.
Understanding results:
Eligibility criteria for testing:
For more information: https://genebygene.com/tryptase/#resources https://genebygene.com/tryptase/#w-tabs-1-data-w-pane-1
The FamilyTreeDNA website of Gene By Gene has an additional feature of the Health report. This report includes:
Upon receiving the report, a free-of-cost, half an hour telemedicine appointment with a consulting physician (or a genetic counselor) is scheduled to give the consumer a proper interpretation of their report.
Xcode Life’s Gene Health Report - 55 Traits, $20 - No DNA test required
| Test | Pricing |
| Court Admissible Paternity Test | $425 |
| Peace of Mind DNA Test (paternity) | $189 |
| Court Admissible Maternity Test | $475 |
| Family Finder | |
| mtFull Sequence | |
| YDNA37 | |
| YDNA111 | |
| Big Y-700 | |
| Tovana Health Report + Family Finder |
The company claims to respect consumer privacy and does not sell or rent out personal information of any kind to a third party without the consumers’ consent.
The company only collects and stores consumer data to operate effectively and provide the best experience of its products. Personal information includes, but is not limited to, names, phone numbers, physical or mailing addresses, email addresses, and genetic test results.
The company uses the stored data to communicate about test progress and results and marketing communications occasionally. One can opt out at any time.
The information is stored in the company headquarters, adjacent to the Genomics Research Center in Houston, Texas.
In addition to Gene By Gene’s Privacy Policy, FamilyTreeDNA adheres to the Genetic Genealogy Guidelines proposed by the Genetic Genealogy Standards Committee in January 2015.
For more information, read Gene By Gene’s privacy policy.
In 2018, in collaboration with Dr. Barbara Rae Venter, Gene By Gene helped law enforcement officials arrest the “Golden State Killer” through its investigative genetic genealogy services.
In January 2021, Gene By Gene went for a merger with Australian genomics firm myDNA headed by Dr. Lior Rauchberger.
| Gene By Gene | Xcode Life | |
| Type of genetic testing | DNA kits | DNA raw data analysis |
| Ancestry reports | Offered (by FTDNA) | Not offered |
| Health report | Offered | Offered |
| Pharmacogenomic report | Not offered | Offered |
| Raw data upload | Only autosomal DNA data from AncestryDNA, 23andMe and MyHeritage | DNA raw data from all major providers accepted. Comprehensive list |
| Number of reports | 2 | 13 |
| Report updates | No | Quarterly updates |
| Price | Tests: | Reports: $10-$40Bundles: $49-$99Price details |
| Free tools | Not available | 3 free tools (Gene Tool, DNA raw data merge, DNA raw data convert) available for all |
| Sample reports | Only for carrier screening and Health report | Available as detailed report walkthrough videos |
NB: Information on Gene By Gene as collected up till 03rd Aug 2021.
